
Looseleaf Study Guide For Chemistry
4th Edition
ISBN: 9781259970214
Author: Julia Burdge
Publisher: MCGRAW-HILL HIGHER EDUCATION
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25.2, Problem 3CP
Identify the name of the following compound:
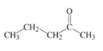
a)
4-Pentanone
b)
2-Pentanone
c)
Ethylpentanal
d)
Propylpentanal
e)
Propylethanal
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the following organic compounds:
a) 2-ethyl-4-methyl-2-pentanol
c) 2-methylpropyl hexanoate
b) 1,3-dimethylbenzene
d) 2-aminoethanoic acid
Write the structural formula for each of the following compounds
2,2-dimethyl-1-butanol
a)
b) 2-methylcyclopentanol
c) 2-phenylethanol
d) 2-methyl-2-propen-1-ol
What is the IUPAC name of the following compound?
a)
2-ethyl-4-pentanol
b)
4-methyl-2-hexanol
c)
4-ethyl-2-pentanol
d)
3-methyl-5-hexanol
Chapter 25 Solutions
Looseleaf Study Guide For Chemistry
Ch. 25.1 - Prob. 1PPACh. 25.1 - Prob. 1PPBCh. 25.1 - Prob. 1PPCCh. 25.2 - Prob. 1PPACh. 25.2 - Prob. 1PPBCh. 25.2 - Prob. 1PPCCh. 25.2 - Prob. 1CPCh. 25.2 - Prob. 2CPCh. 25.2 - Identify the name of the following compound: a)...Ch. 25.2 - Prob. 4CP
Ch. 25.2 - Prob. 5CPCh. 25.2 - Prob. 6CPCh. 25.3 - Prob. 1PPACh. 25.3 - Prob. 1PPBCh. 25.3 - Prob. 1PPCCh. 25.3 - Prob. 1CPCh. 25.3 - Prob. 2CPCh. 25.3 - Prob. 3CPCh. 25.3 - Prob. 4CPCh. 25.4 - Prob. 1PPACh. 25.4 - Prob. 1PPBCh. 25.4 - Prob. 1PPCCh. 25.5 - Prob. 1PPACh. 25.5 - Prob. 1PPBCh. 25.5 - Prob. 1PPCCh. 25.5 - Prob. 1CPCh. 25.5 - Prob. 2CPCh. 25 - Prob. 1QPCh. 25 - 25.2 Why was Wöhler’s synthesis of urea so...Ch. 25 - Prob. 3QPCh. 25 - Prob. 4QPCh. 25 - Prob. 5QPCh. 25 - Prob. 6QPCh. 25 - Prob. 7QPCh. 25 - Prob. 8QPCh. 25 - Prob. 9QPCh. 25 - Prob. 10QPCh. 25 - Prob. 11QPCh. 25 - Prob. 12QPCh. 25 - Prob. 13QPCh. 25 - Prob. 14QPCh. 25 - Prob. 15QPCh. 25 - Identify the functional groups in the...Ch. 25 - Prob. 17QPCh. 25 - Prob. 18QPCh. 25 - Prob. 19QPCh. 25 - Prob. 20QPCh. 25 - Prob. 21QPCh. 25 - Prob. 22QPCh. 25 - Prob. 23QPCh. 25 - Prob. 24QPCh. 25 - Prob. 25QPCh. 25 - Prob. 26QPCh. 25 - Prob. 27QPCh. 25 - Prob. 28QPCh. 25 - Prob. 29QPCh. 25 - Prob. 30QPCh. 25 - Prob. 31QPCh. 25 - Prob. 32QPCh. 25 - Prob. 33QPCh. 25 - Prob. 34QPCh. 25 - Prob. 35QPCh. 25 - Prob. 36QPCh. 25 - Prob. 37QPCh. 25 - Prob. 38QPCh. 25 - Prob. 39QPCh. 25 - Prob. 40QPCh. 25 - Prob. 41QPCh. 25 - Prob. 42QPCh. 25 - Prob. 43QPCh. 25 - Prob. 44QPCh. 25 - Prob. 45QPCh. 25 - Prob. 46QPCh. 25 - Prob. 47QPCh. 25 - Prob. 48QPCh. 25 - Prob. 49QPCh. 25 - Prob. 50QPCh. 25 - Prob. 51QPCh. 25 - Prob. 52QPCh. 25 - Prob. 53QPCh. 25 - Prob. 54QPCh. 25 - Prob. 55QPCh. 25 - Prob. 56QPCh. 25 - Prob. 57QPCh. 25 - Prob. 58QPCh. 25 - Prob. 59QPCh. 25 - Prob. 60QPCh. 25 - Prob. 61QPCh. 25 - Prob. 62QPCh. 25 - Prob. 63QPCh. 25 - Prob. 64QPCh. 25 - Prob. 65QPCh. 25 - Prob. 66QPCh. 25 - Prob. 67QPCh. 25 - Prob. 68QPCh. 25 - Prob. 69QPCh. 25 - Prob. 70QPCh. 25 - Prob. 71QPCh. 25 - Prob. 72QPCh. 25 - Prob. 73QPCh. 25 - Prob. 74QPCh. 25 - Prob. 75QPCh. 25 - Prob. 76QPCh. 25 - Prob. 77APCh. 25 - Prob. 78APCh. 25 - Prob. 79APCh. 25 - Prob. 80APCh. 25 - Prob. 81APCh. 25 - Match each molecular model with the correct...Ch. 25 - Prob. 83APCh. 25 - Prob. 84APCh. 25 - Prob. 85APCh. 25 - Prob. 86APCh. 25 - Prob. 87APCh. 25 - Prob. 88APCh. 25 - Prob. 89APCh. 25 - Prob. 90APCh. 25 - Prob. 91APCh. 25 - Prob. 92APCh. 25 - Prob. 93APCh. 25 - Prob. 94APCh. 25 - Prob. 95APCh. 25 - Prob. 96APCh. 25 - Prob. 97APCh. 25 - Prob. 98APCh. 25 - Prob. 99APCh. 25 - Prob. 100APCh. 25 - All alkanes give off heat when burned in air. Such...Ch. 25 - Prob. 102APCh. 25 - Prob. 1SEPPCh. 25 - Prob. 2SEPPCh. 25 - Prob. 3SEPPCh. 25 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Question 39 of 50 Submit What is the correct IUPAC name for the compound shown here? A) 2-butanol B) 3-butanol C) 2-pentanol НО D) 2-hydroxybutane E) 2-butanalarrow_forward47) What is the common name of this compound? n CH3-C-O-CH3 A) ethyl methyl ester B) diethyl ester C) ethyl methanoate D) 2-ether-2-butanone E) ethyl acetatearrow_forwardWhat is the line structure and the functional groups present (if any)? a) cyclohexane b) cyclohexanol c) cyclohexanonearrow_forward
- Q1: What is the IUPAC name of the organic compound? Q2: What is the IUPAC name of the organic compound?arrow_forwardDraw the following condensed structural diagrams b) diethyl ketone c) N,N-dimethyl-2-propanamine d) 3-methyl-5-chloroheptanal e) 3-methoxyhexane f) methyl pentanoatearrow_forwardDraw the structural formulas for the following hydrocarbon molecules: d) 2-methyl-3-hexene e) 3-ethyl-1-hexyne f) 1,3-diethylbenzenearrow_forward
- Which of the following is the IUPAC name of sec-butyl alcohol? a) 1-butanol b) 2-butanol c) 2-methyl-1-propanol d) 2-methyl-2-propanolarrow_forwardDraw the following compounds: b) pent-3-en-1-ol e) 1,2-dibromobenzene a) 3-ethyl-2,4-dimethylhept-1-ene c) N-ethylhexan-3-aminearrow_forwardWhat is the correct IUPAC name for the compound shown here? A) 2-butanol B) 3-butanol C) 2-pentanol Но D) 2-hydroxybutane E) 2-butanalarrow_forward
- Draw the following organic compounds explicit formulas. a) 2-isobutyl-3-methyl-cyclohexanol b) 2-methoxy-2-methylpropanearrow_forwardWhat is different between: n-octanol and octanol?arrow_forwardWhat is the classification of the compound shown here? H A) ester B) carboxylic acid C) ketone D) alcohol E) aldehydearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY