
Concept explainers
Interpretation:
The isoprene unit in
Concept introduction:
The terpenes are the repeating isoprene units.
The isoprene unit is nothing but the carbon skeleton of isoprene without double bonds.
The isoprene molecule has head and tail. The structure of isoprene
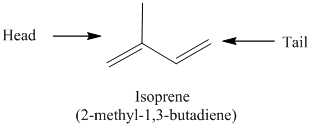
The isoprene units are together linked in head-to-tail configuration. The tail of the first isoprene molecule joins with the head of the second isoprene molecule.
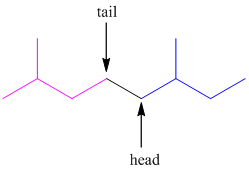
In higher terpenes, tail-to-tail linkage can also occur. The tail of the first isoprene molecule joins with the tail of the second isoprene molecule.
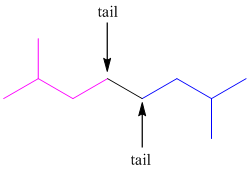
As the terpenes forms by isomerization of isoprene unit, both the double bonds of isoprene may not be present in the structure of terpenes.
On the basis of number of isoprene units
Want to see the full answer?
Check out a sample textbook solution
Chapter 25 Solutions
ORGANIC CHEMISTRY (LL)-W/SOLN.>CUSTOM<
- The molar heat of combustion of gaseous cyclopropane is -2089 Kj/mol, that for gaseous cyclopentene is -3317 Kj/mol. Calculate the heat of combustion per CH2 group in the two cases and account for the differencearrow_forwardExcluding compounds that contain methyl or ethyl groups, write structural formulas for all the bicyclic isomers of (a) C5H8 and (b) C6H10.arrow_forwardAnalyze the following structure of limonene in terms of isoprene units joined by head-to-tail linkages. (1) How many isoprene units are there in a limonene molecule? (2) Classify limonene as a monoterpene (one terpene unit), a sesquiterpene (1.5 terpene units), or a diterpene (2 terpene units).arrow_forward
- (a) Locate the isoprene units in lycopene, the red pigment in tomatoes. (b) Which isoprene units are connected in a head-to-tail fashion? (c) Label any other isoprene unit as connected in a head-tohead fashion or a tail-to-tail fashion. (d) Classify lycopene as a monoterpene, sesquiterpene, and so on.arrow_forwardThe molar heat of combustion of gaseous cyclopropaneis -2089 kJ/mol; that for gaseous cyclopentane is-3317 kJ/mol. Calculate the heat of combustion per CH2group in the two cases, and account for the difference.arrow_forwardThe molar heat of combustion of gaseous cyclopropane is -2089 kJ/mol; that for gaseous cyclopentane is -3317 kJ/mol. Calculate the heat of combustion per CH2 group in the two cases, and account for the difference.arrow_forward
- 8. Give three structural formulas each of acyclic and carbocyclic compounds having the composition 1) C5H8; 2) C5H100. 16:47 /arrow_forwardWhy are carbon atoms 1 and 3 of glyceraldehyde not considered chiral?arrow_forwardWhen compound CH3-C(CH3)=CH-CH2-CH(CH3)2 is the SUBSTRATE (reactant) in a HALOGENATION reaction with Br2 (and CH2Cl2 as the solvent), a correct condensed structural formula for the MAJOR SUBSTRATE PRODUCT of this reaction is:arrow_forward
- Q.2. Draw the structure for trans-3-chloro-2-isopropyl-4-hexen-1-ol.arrow_forwardRecognize fused aromatic systems such as polynuclear aromatic hydrocarbons andfused heterocyclic compounds, and use the theory of aromatic compounds to explaintheir properties.arrow_forwardName all functional groups in the compound and what type of aliphatic it is. CH;=CH-CH2- -CH, alkane; carboxylic acid alkene; ester alkane; ketone alkene; aldehydearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





