
(a)
Interpretation:
The structure of
Concept introduction:
The systematic naming of organic compound is given by
Rules for writing IUPAC name from the structural formula are given below.
- First, identify the longest carbon chain.
- The next step is to identify the groups attached to the longest chain.
- Identify the position, location, and a number of the substituents bonded to the carbon chain.
- Use prefix di, tri, tetra if the same type of substituents is present.
- Name the substituents in alphabetical order.
- In the heterocyclic compound, azo for nitrogen, oxo for oxygen, thio for sulfur and so on is used.
Answer to Problem 26.1P
The structure of the given compound,
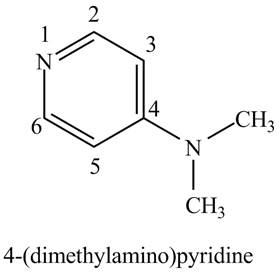
Explanation of Solution
The name of the given compound
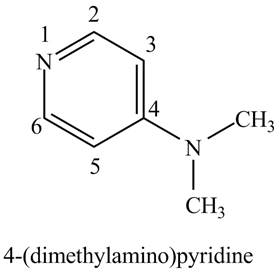
Figure 1
The structure of the given compound,
(b)
Interpretation:
The structure of
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from the structural formula are given below.
- First, identify the longest carbon chain.
- The next step is to identify the groups attached to the longest chain.
- Identify the position, location, and a number of the substituents bonded to the carbon chain.
- Use prefix di, tri, tetra if the same type of substituents is present.
- Name the substituents in alphabetical order.
- In the heterocyclic compound, azo for nitrogen, oxo for oxygen, thio for sulfur and so on is used.
Answer to Problem 26.1P
The structure of the given compound,
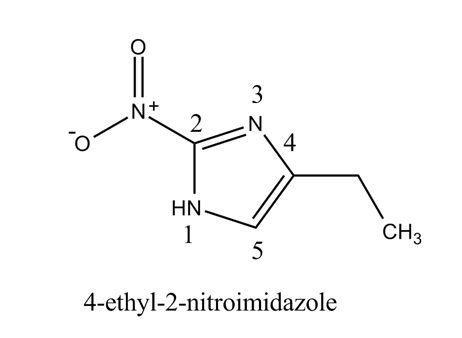
Explanation of Solution
The given compound

Figure 2
The structure of the given compound,
Want to see more full solutions like this?
Chapter 26 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- Give the chemical tests to distinguish between the following pairs of compounds :(i) Methylamine and Dimethylamine(ii) Aniline and N-methylanilinearrow_forwardDraw the structures of the following compounds:(a) tert-butylaminearrow_forward(a) Compound Z is a tertiary aromatic amine with the formula, C8H11N. Provide a chemical structure for compound Z. (b)nDraw the structure of the product formed exclusively when nitrous acid reacts with Z.arrow_forward
- Draw structures corresponding to the following names:(a) 3,4-Dimethylhexanoic acid(b) Phenylacetic acid(c) 3,4-Dinitrobenzoic acid(d) Triethylammonium butanoatearrow_forwardGive the chemical tests to distinguish between the following pairs of compounds :(i) Ethyl amine and Aniline(ii) Aniline and Benzylaminearrow_forwardIn each of the following reactions, two possible organic products can be formed. Draw both organic products in each case and then circle the one formed in greatest quantity in each case. HC (a) 1) NaH, 2) acid (b) CH,CH,OH (c) CH,CH,OH NH2 (d) Oarrow_forward
- 34) The product formed by the reaction of toluene with chlorine in the presence of sunlight is: (a) o-chlorotoluene (b) 2,4-dichlorotoluene (c) p-chlorotoluene (d) Benzylchloridearrow_forwardName and draw a structural formula for the major product of each alkene addition reaction: CH S (a) CH₂C=CH₂ + HI →→→→ (b) CH 3 + HClarrow_forwardGive the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forward
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardKetene, H2C=C=O, is an important industrial chemical. Predict the products that would be formed when ketene reacts with **hint: Markovnikov addition occurs. (a) ethanol (b) acetic acid (c) ethylamine.arrow_forwardDraw the structure for the following compounds:(i) Ethanoic acid(ii) Bromopentane(iii) Butane(iv) Hexanalarrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
