
Concept explainers
(a)
Interpretation:
The product formed when
Concept Introduction:
Pyridine is a heterocyclic compound which contains nitrogen atom. Pyridine nitrogen atom contain a lone pair. The lone pair of pyridine is not involved in the resonance with the
Answer to Problem 26.27AP
The product formed when
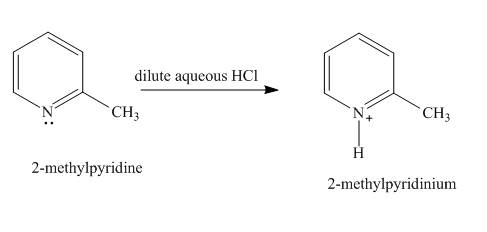
Explanation of Solution
In the given compound, the lone pair of the pyridine nitrogen is not involved in the resonance with the ring. Pyridine acts as base in the presence of acid. The nitrogen abstracts the proton from acid. The product formed is

Figure 1
The product formed in the given reaction is
(b)
Interpretation:
The product formed when
Concept Introduction:
Pyridine is a heterocyclic compound which contains nitrogen atom. Pyridine nitrogen atom contain a lone pair. The lone pair of pyridine is not involved in the resonance with the aromatic ring. No delocation of electron takes place, hence pyridine acts as a base. The hydrogen atoms of pyridine ring are not highly acidic, so mild base can’t abstract proton from pyridine. It requires a strong base for abstraction of proton.
Answer to Problem 26.27AP
No reaction takes place when
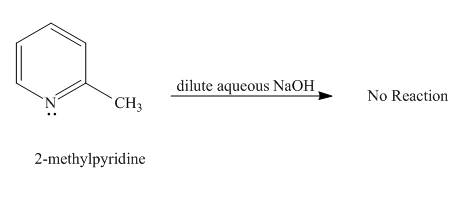
Explanation of Solution
Protons of pyridine are not highly acidic in nature. Sodium hydroxide cannot abstract proton from pyridine. It requires a very strong base for the removal of proton from pyridine to takes place. Hence, no reaction takes place between

Figure 2
No product is formed in the given reaction.
(c)
Interpretation:
The product formed when
Concept Introduction:
Pyridine is a heterocyclic compound which contains nitrogen atom. Pyridine nitrogen atom contain a lone pair. The lone pair of pyridine is not involved in the resonance with the aromatic ring. No delocation of electron takes place, hence pyridine acts as a base. The hydrogen atoms of pyridine ring are not highly acidic, so mild base can’t abstract proton from pyridine. It requires a strong base for abstraction of proton.
Answer to Problem 26.27AP
The product formed when
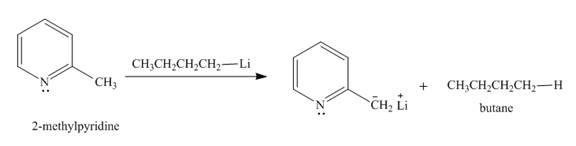
Explanation of Solution
The butyl-lithium compound is a very strong base. It will abstract proton from the methyl group of
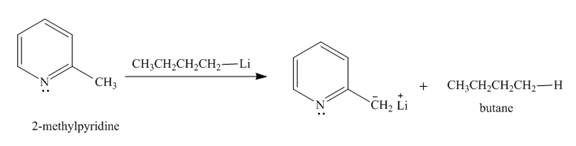
Figure 3
The product formed in the given reaction is
(d)
Interpretation:
The product formed when
Concept Introduction:
Nitration reaction is aromatic electrophilic substitution reaction. The nitrating mixture contains nitric acid and sulfuric acid. The nitrosonium ion is formed as electrophile. The methyl group is an activating group, so it promotes electrophilic substitution reaction at ortho-para position.
Answer to Problem 26.27AP
The product formed when
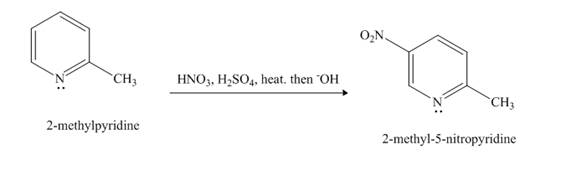
Explanation of Solution
The compound

Figure 4
The product formed in the given reaction is
(e)
Interpretation:
The product formed when
Concept Introduction:
Pyridine is a heterocyclic compound which contains nitrogen atom. Pyridine nitrogen atom contain a lone pair. The lone pair of pyridine is not involved in the resonance with the aromatic ring. Hydrogen peroxide is a strong oxidizing agent. It will oxidize the nitrogen atom of pyridine to form
Answer to Problem 26.27AP
The product formed when
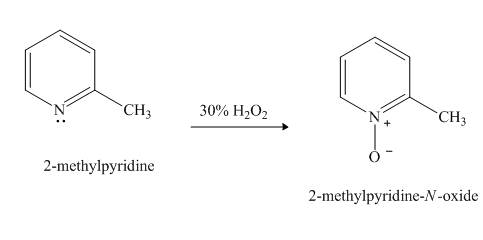
Explanation of Solution
Hydrogen peroxide is an oxidizing agent. It will oxidize the given compound. The oxidation reaction takes place at the nitrogen atom.

Figure 5
The product formed in the given reaction of
(f)
Interpretation:
The product formed when
Concept Introduction:
Pyridine is a heterocyclic compound which contains nitrogen atom. Pyridine nitrogen atom contain a lone pair. The lone pair of pyridine is not involved in the resonance with the aromatic ring. No delocation of electron takes place, hence pyridine acts as a base. The nitrogen atom lone pair attacks the methyl group of methyl iodide.
Answer to Problem 26.27AP
The product formed when
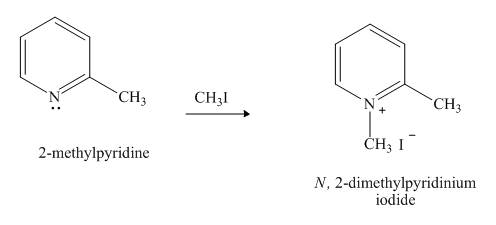
Explanation of Solution
The lone pair of nitrogen atom of pyridine is not involved in the resonance with the aromatic ring. The lone pair makes the pyridine compound basic in nature. When compound

Figure 6
The product formed in the given reaction of
(g)
Interpretation:
The product formed when
Concept Introduction:
Pyridine is a heterocyclic compound which contains nitrogen atom. Pyridine nitrogen atom contain a lone pair. The lone pair of pyridine is not involved in the resonance with the aromatic ring. No delocation of electron takes place, hence pyridine acts as a base. Lithiated pyridine compound reacts with
Answer to Problem 26.27AP
The product formed when
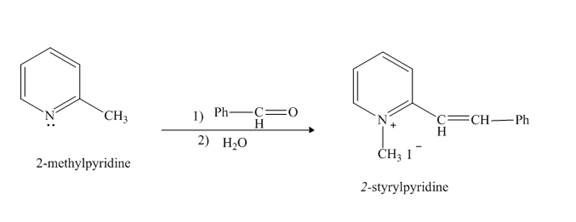
Explanation of Solution
The lithiation product formed reacts with benzaldehyde, nucleophilic addition reaction takes place to form

Figure 7
The product formed when
(h)
Interpretation:
The product formed when
Concept Introduction:
Pyridine is a heterocyclic compound which contains nitrogen atom. Pyridine nitrogen atom contain a lone pair. The lone pair of pyridine is not involved in the resonance with the aromatic ring. Hydrogen peroxide is a strong oxidizing agent. It will oxidize the nitrogen atom of pyridine to form
Answer to Problem 26.27AP
The product formed when
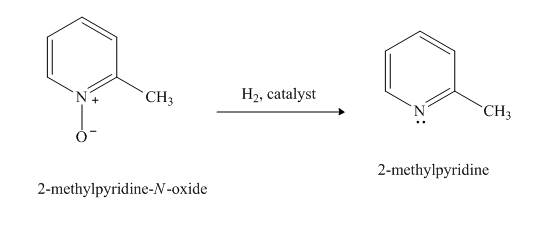
Explanation of Solution
The compound

Figure 8
The product formed when
Want to see more full solutions like this?
Chapter 26 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- (a) A hydrocarbon isolated from fish oil and from plankton was identified as 2,6,10,14-tetramethyl-2-pentadecene. Write its structure.(b) Alkyl isothiocyanates are compounds of the type RN C S. Write a structural formula for allyl isothiocyanate, a pungent-smelling compound isolated from mustard.(c) Grandisol is one component of the sex attractant of the boll weevil. Write a structural formula for grandisol given that R in the structure shown is an isopropenyl group.arrow_forwardPropose structures for molecules that fit the following descriptions:(a) An aldehyde with the formula C5H10O(b) An ester with the formula C6H12O2(c) A compound with the formula C3H7NOS that is both anamide and a thiolarrow_forwardDraw the structural formulas of the following compounds:(a) 2,3-Dimethylpentanal(b) 1,3-Dibromopropanone(c) 4-hydroxy-4-methylhexan-2-onearrow_forward
- 8arrow_forward(a) Draw the three isomers of benzenedicarboxylic acid.(b) The isomers have melting points of 210 °C, 343 °C, and 427 °C. Nitration of the isomers at all possible positions was once used to determine their structures. The isomer that melts at 210 °C gives two mononitro isomers. The isomer that melts at 343 °C gives three mononitro isomers. The isomer that melts at 427 °C gives only one mononitro isomer. Show which isomer has which melting point.arrow_forward(b) 3-methyl-2-butanol reacts with concentrated sulphuric acid to form 2-methyl-2- butene. Write the mechanism for the reaction.arrow_forward
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardPredict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.(a) PhMgBr, then H3O+ (b) Tollens reagent (c) semicarbazide and weak acid(d) excess ethanol and acid (e) propane-1,3-diol, H+ (f) zinc amalgam and dilute hydrochloric acidarrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) сон COOH ОН ОН COOH COOH KMnO4 H2Oarrow_forward
- 6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forwardDraw structures corresponding to the following names: (a) 3-hydroxyhexanoic acid (b) 2-iodo-2-methyloctanoic acid (c) 2-butynoic acid (d) 5-ethyl-6-oxoheptanoic acid (e) o-Hydroxybenzoic acid (f) cis-3-isopropylcyclohexanecarboxylic acidarrow_forwardDraw structural formulas for these ketones. (a) Ethyl isopropyl ketone (b) 2-Chlorocyclohexanone (c) 2,4-Dimethyl-3-pentanone (d) Diisopropyl ketone (e) Acetone (f) 2,5-Dimethylcyclohexanonearrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

