
Concept explainers
Devise a synthesis of each substituted cyclopropane. Use acetylene
a. ![]() b.
b.  c.
c. ![]() d.
d. 
+enantiomer +enantiomer
(a)
Interpretation: A synthesis of given substituted cyclopropane from acetylene
Concept introduction: The nonhalogenated cyclopropanes are synthesized by the treatment of an alkene with
Answer to Problem 26.44P
A synthesis of given substituted cyclopropane from acetylene
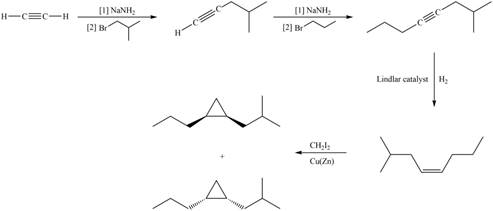
Explanation of Solution
The synthesis of given compound involves four steps.
The first step involves reaction of acetylene with
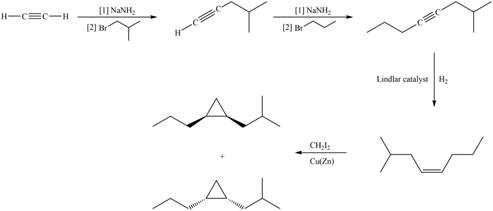
Figure 1
A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents, is shown in Figure 1.
(b)
Interpretation: A synthesis of given substituted cyclopropane from acetylene
Concept introduction: The nonhalogenated cyclopropanes are synthesized by the treatment of an alkene with
Answer to Problem 26.44P
A synthesis of given substituted cyclopropane from acetylene
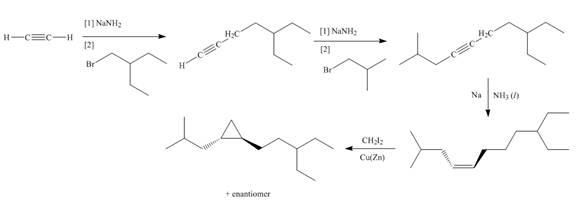
Explanation of Solution
The synthesis of given compound involves four steps.
The first step involves reaction of acetylene with
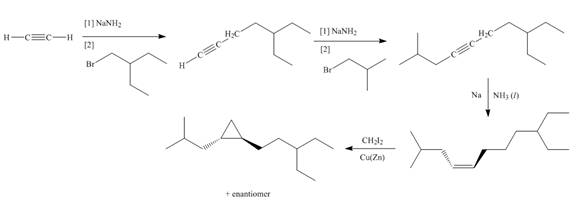
Figure 2
A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents, is shown in Figure 2.
(c)
Interpretation: A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents, is to be stated.
Concept introduction: The nonhalogenated cyclopropanes are synthesized by the treatment of an alkene with
Answer to Problem 26.44P
A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents is,
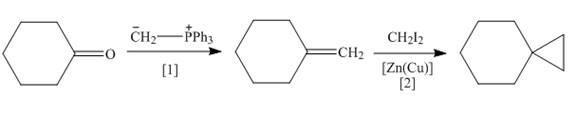
Explanation of Solution
The synthesis of given compound involves two steps.
In the first step of reaction, cyclohexanone undergoes Wittig reaction with Ylide,
These steps are shown below.

Figure 3
A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents, is shown in Figure 3.
(d)
Interpretation: A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents, is to be stated.
Concept introduction: The treatment of
Answer to Problem 26.44P
A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents is,
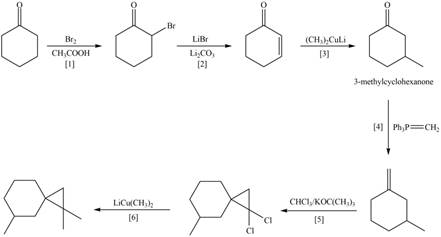
Explanation of Solution
The synthesis of given compound involves six steps.
The first step is alpha bromination of cyclohexanone in the presence of acetic acid. The second part is reaction of
These steps are shown below.
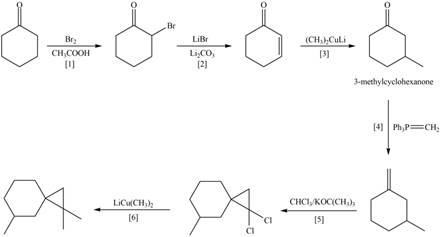
Figure 4
A synthesis of given substituted cyclopropane from cyclohexanone, any organic compounds, and reagents, is shown in Figure 4.
Want to see more full solutions like this?
Chapter 26 Solutions
ALEKS 360 CHEMISTRY ACCESS
- Devise a synthesis of attached alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents.arrow_forwardDevise a synthesis of each compound using 1-bromobutane(CH3CH2CH2CH2Br) as the only organic starting material. You may useany other inorganic reagents.arrow_forwardA key step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involved the reaction of compound A and dihalide B with two equivalents of LDA to form C. Draw a stepwise mechanism for this reaction. β-Vetivone contains a spiro ring system—that is, two rings that share a single carbon atom.arrow_forward
- Sulfur ylides, like the phosphorus ylides, are usefulintermediates in organic synthesis. Methyl trans-chrysanthemate, anintermediate in the synthesis of the insecticide pyrethrin I,can be prepared from diene A and a sulfur ylide. Draw a stepwisemechanism for this reaction.arrow_forwardDevise a synthesis of each compound from cyclohex-2-enone and organic halides having one or two carbons. You may use any other required inorganic reagents.arrow_forward(a) Give an acceptable name for each compound, (b) Draw the organic products formed when A or B is treated with each reagent: [1] H3O+; [2] −OH, H2O; [3] CH3CH2CH2MgBr (excess), then H2O; [4] LiAlH4, then H2O.arrow_forward
- Devise a synthesis of each alkyne. You may use acetylene, benzene, organic halides, ethylene oxide, and any other required inorganic reagents.arrow_forwardDraw a stepwise mechanism for the following substitution. Explain why 2-chloropyridine reacts faster than chlorobenzene in this type of reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
