
Concept explainers
Biaryls, compounds containing two
a.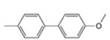 b.
b. 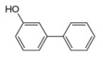 c.
c. 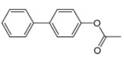
(a)
Interpretation: The synthesis of given biaryl compound using benzene as the starting material and any required organic or inorganic reagents is to be stated, and if more than one route is possible, then both routes are to be drawn.
Concept introduction: The treatment of an organic halide
Answer to Problem 26.47P
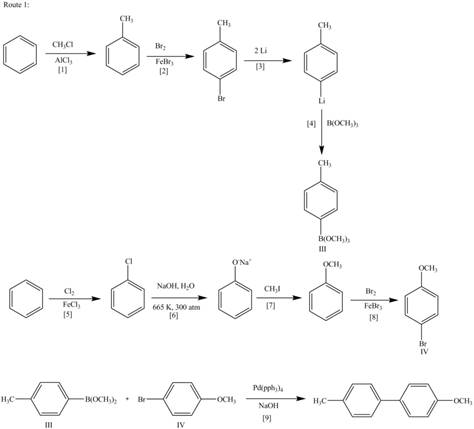
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown below.
Explanation of Solution
The treatment of an organic halide
The retrosynthetic analysis of the given compound is,
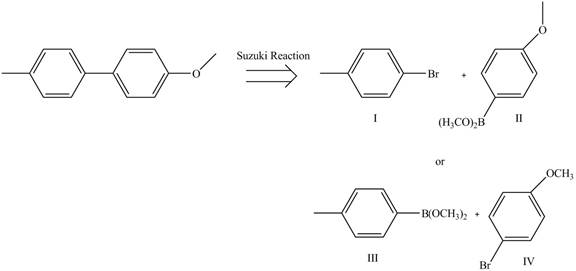
Figure 1
The above analysis shows that there are two possible routes to synthesize the given compound.
Route 1:
The Route 1 involves formation of desired biaryl in nine steps. The first step is Friedel-Crafts alkylation of benzene with
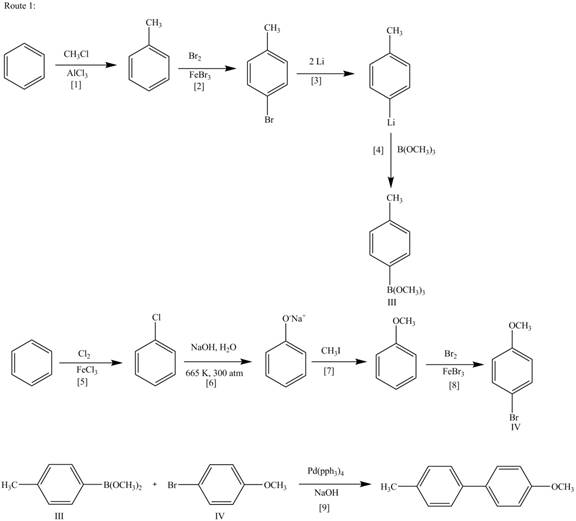
Figure 2
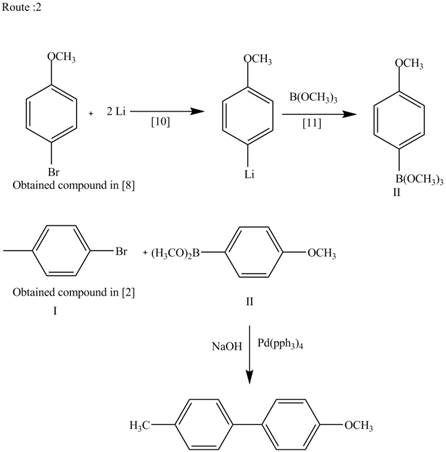
Route 2:
The Route 2 involves
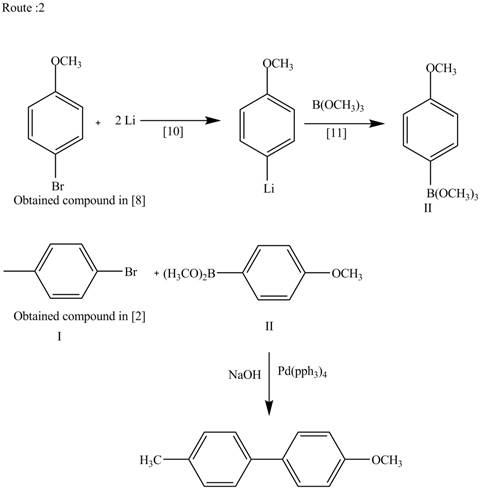
Figure 3
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown in Figure 2, and Figure 3.
(b)
Interpretation: The synthesis of given biaryl compound using benzene as the starting material and any required organic or inorganic reagents is to be stated, and if more than one route is possible, then both routes are to be drawn.
Concept introduction: The treatment of an organic halide
Answer to Problem 26.47P
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown below.
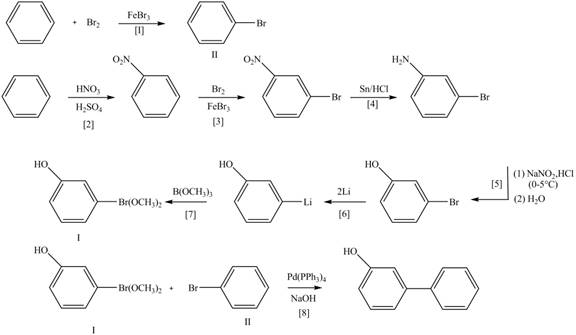
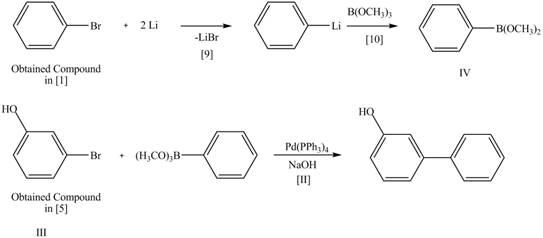
Explanation of Solution
The treatment of an organic halide
The retrosynthetic analysis of the given compound is,
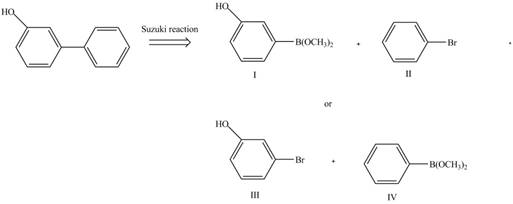
Figure 4
The above analysis shows that there are two possible routes to synthesize the given compound.
Route 1:
The Route 1 involves eight steps. The first step is bromination of benzene in the presence of
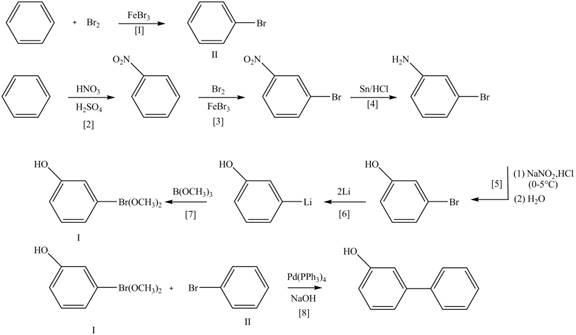
Figure 5
Route 2:
The Route 2 involves
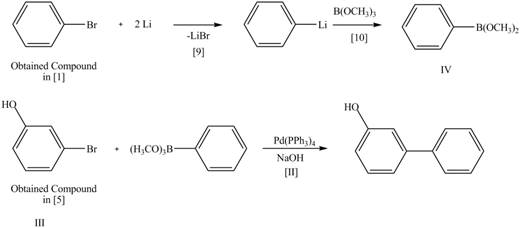
Figure 6
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown in Figure 5, and Figure 6.
(c)
Interpretation: The synthesis of given biaryl compound using benzene as the starting material and any required organic or inorganic reagents is to be stated, and if more than one route is possible, then both routes are to be drawn.
Concept introduction: The treatment of an organic halide
Answer to Problem 26.47P
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown below.
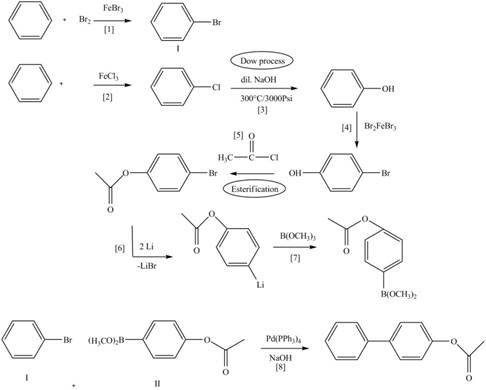
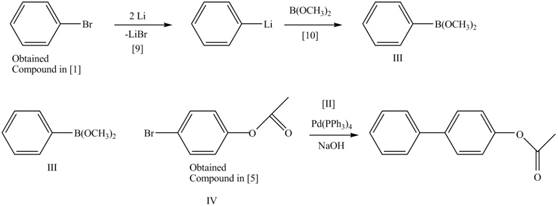
Explanation of Solution
The treatment of an organic halide
The retrosynthetic analysis of the given compound is,
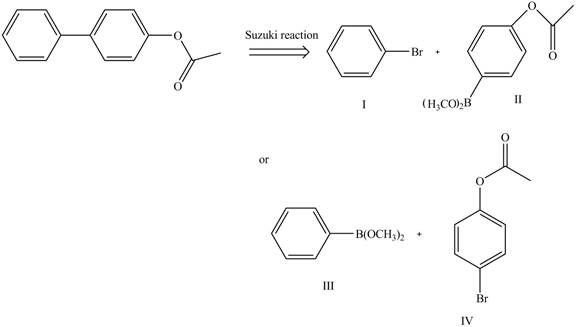
Figure 7
The above analysis shows that there are two possible routes to synthesize the given compound.
Route 1:
The Route 1 involves eight steps. The first step is bromination of benzene in the presence of
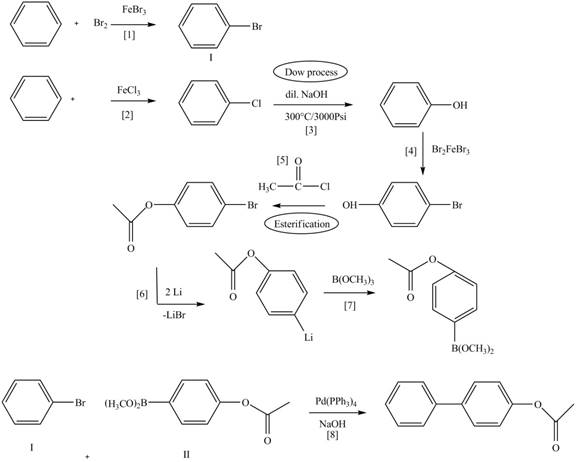
Figure 8
Route 2:
The Route 2 involves
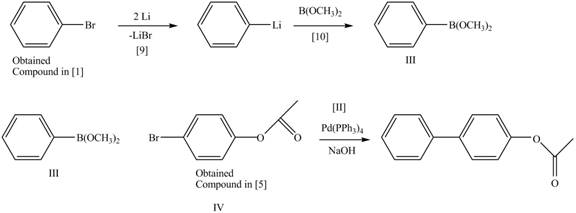
Figure 9
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown in Figure 8, and Figure 9.
Want to see more full solutions like this?
Chapter 26 Solutions
ORGANIC CHEMISTRY-STUDY GUIDE PACKAGE
- Starting with benzene and using any other necessary reagents of your choice, create a synthesis (or sytheses) for each of the following compounds.arrow_forwardOrganometallic compounds act as nucleophiles under very specific conditions, and can generate alcohols, alkanes, among others. Describe in detail what these reaction conditions are so that organometallics can perform the addition reaction to carbonyl.arrow_forwardFor each horizontal row of substituted benzenes, indicatea. the one that is the most reactive in an electrophilic aromatic substitution reaction.b. the one that is the least reactive in an electrophilic aromatic substitution reaction.c. the one that yields the highest percentage of meta product in an electrophilic aromatic substitution reaction.arrow_forward
- Provide the major substitution product for the following reaction.arrow_forwardChoose one base, and conditions for the reaction to include temperature (low heat vs high heat) and specific solvent would be most suitable to promote the following alkyl halide to undergo an E2 mechanism. Draw the mechanism including hydride/alkyl shifts, intermediates, and ALL transition states as applicable. Draw products indicating the regiochemistry indicating the Zaitsev/Hoffmann products as well as stating which will be the major and which the minor product. Base: Solvent: Temperature: Mechanism:arrow_forwardChemistry Please help explain this textbook question: Although N, N -dimethylaniline is extremely reactive toward electrophilic aromatic substitution and is readily substituted by weak electrophiles, such as diazonium and nitronium ions, this reactivity is greatly diminished by the introduction of an alkyl substituent in an ortho position.arrow_forward
- Propose a synthetic route that would synthesize the substituted benzenes as a major product. You can only use the starting material indicated, but you can use any other additional solvents and inorganic reagents as needed.?arrow_forwardFor each of the questions below – determine whether the following combinations of reactants, will react by substitution (SN1 or SN2 mechanism), elimination (E1 or E2 mechanism) or will not react under the conditions given. draw the final structure and explain the logic for each decision. There may be multiple possible options for some reactions. You must include all 4 aspects of these reactions, substrate, nucleophile, solvent and leaving group in the decisionarrow_forwardProvide the appropriate reagent(s) for the following transformationarrow_forward
- Write the products for (A-I). Label products with the mechanism (Sn2, Sn1, E1, E2) that produced it and write out and label major vs minor elimination products.arrow_forwardWhich of the following is a correct statement regarding electrophilic aromatic substitution? A. Many suitable electrophiles are unreactive and can be stored for long periods of time prior to use. B. The carbocation intermediate will lose a proton to regain aromaticity, usually from a position other than the site of the electrophilic attack. C. Formation of the carbocation intermediate has a high activation barrier due to loss of aromaticity. D. Reformation of the aromatic ring has a low activation barrier and therefore occurs slowly. E. The carbocation intermediate has several resonance structures and is negatively charged.arrow_forwardIn a Wittig reaction, a ketone or aldehyde reacts as an electrophile with a nucleophile called a Wittig reagent (or phosphonium ylide) to produce an alkene. The Wittig reagent is commonly synthesized first in a two-step process beginning with an alkyl halide, then reacted with the carbonyl compound. In this problem, you'll explore the mechanism of a multi-step synthesis to make an alkene using the Wittig approach.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

