
EBK GENERAL CHEMISTRY
11th Edition
ISBN: 9780134419022
Author: Bissonnette
Publisher: PEARSON CUSTOM PUB.(CONSIGNMENT)
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 26, Problem 89IAE
Levomethadyl acetate (shown below) is used in the treatment of narcotic addiction.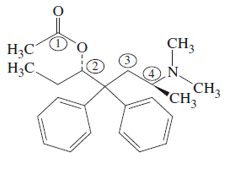
a. Name the
b. What is the hybndization of the numbered carbon atoms and the nitrogen atom?
c. Which, if any, of the numbered carbon atoms are chiral?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1.
Which compounds contain chiral centers? Show your solution for each
ww ww w w
item.
a. 2-Chloropentane
b. 3-Chloro-1-pentene
c. 3-Chloropentane
d. 1,2-Dichloropropane
28. A structural feature with in a molecule
which is responsible for chirality is called
c. chirality
d. Chiral C
e. Chiral center
d. all
29. Optically active compounds have isomer
a Two
C. four
b. Three
d. five
30. Propanone and propenol are
a. structural isomers
C. tautomer
b. metamers
d position isomer
Draw the ringed structure for D- Glucose
Chapter 26 Solutions
EBK GENERAL CHEMISTRY
Ch. 26 - Prob. 1ECh. 26 - Draw a structural formula for each of the...Ch. 26 - Prob. 3ECh. 26 - Write structural formulas corresponding to these...Ch. 26 - Prob. 5ECh. 26 - Prob. 6ECh. 26 - Prob. 7ECh. 26 - Prob. 8ECh. 26 - What is the relationship, if any, between the...Ch. 26 - Prob. 10E
Ch. 26 - Prob. 11ECh. 26 - Prob. 12ECh. 26 - Identify the chiral carbon atoms, ¡f any, in the...Ch. 26 - Prob. 14ECh. 26 - Identify the chiral carbon atoms, ¡f any, in the...Ch. 26 - Prob. 16ECh. 26 - Prob. 17ECh. 26 - Prob. 18ECh. 26 - Prob. 19ECh. 26 - By name or formula, give one example of each of...Ch. 26 - Prob. 21ECh. 26 - Prob. 22ECh. 26 - Prob. 23ECh. 26 - Prob. 24ECh. 26 - Prob. 25ECh. 26 - Prob. 26ECh. 26 - Prob. 27ECh. 26 - Prob. 28ECh. 26 - Prob. 29ECh. 26 - Prob. 30ECh. 26 - Prob. 31ECh. 26 - Prob. 32ECh. 26 - Prob. 33ECh. 26 - Prob. 34ECh. 26 - Does each of the following names convey sufficient...Ch. 26 - Prob. 36ECh. 26 - Prob. 37ECh. 26 - Supply condensed structural formulas for the...Ch. 26 - Prob. 39ECh. 26 - Prob. 40ECh. 26 - Classify the carbon atoms in, a. methylbutane, and...Ch. 26 - Classity the carbon atoms in a....Ch. 26 - Prob. 43ECh. 26 - Draw Newman projections for the staggered and...Ch. 26 - Draw the most stable conformation for the molecule...Ch. 26 - Prob. 46ECh. 26 - Prob. 47ECh. 26 - Prob. 48ECh. 26 - Prob. 49ECh. 26 - Prob. 50ECh. 26 - Prob. 51ECh. 26 - Prob. 52ECh. 26 - Prob. 53ECh. 26 - Prob. 54ECh. 26 - Prob. 55ECh. 26 - Prob. 56ECh. 26 - Draw suitable structural formulas to show that...Ch. 26 - Which of the following pairs of molecules are...Ch. 26 - Prob. 59ECh. 26 - Prob. 60ECh. 26 - Name the following molecules with the appropriate...Ch. 26 - Name the following molecules with the appropriate...Ch. 26 - Name the following molecules with the appropriate...Ch. 26 - Prob. 64ECh. 26 - Draw the structure for each of the following. a....Ch. 26 - Prob. 66ECh. 26 - Prob. 67ECh. 26 - Prob. 68ECh. 26 - Prob. 69ECh. 26 - Prob. 70ECh. 26 - Prob. 71ECh. 26 - Prob. 72ECh. 26 - Prob. 73ECh. 26 - Prob. 74ECh. 26 - Supply condensed or structural formulas for the...Ch. 26 - Prob. 76IAECh. 26 - Prob. 77IAECh. 26 - Prob. 78IAECh. 26 - Prob. 79IAECh. 26 - Prob. 80IAECh. 26 - Combustion of a 0.1908 g sample of a compound gave...Ch. 26 - Prob. 82IAECh. 26 - In the monochiorination of hydrocarbons, a...Ch. 26 - A particular colorless organic liquid is known to...Ch. 26 - Prob. 85IAECh. 26 - Give the systematic names, including any...Ch. 26 - Prob. 87IAECh. 26 - Prob. 88IAECh. 26 - Levomethadyl acetate (shown below) is used in the...Ch. 26 - Thiamphenicol (shown below) is an antibacterial...Ch. 26 - Prob. 91IAECh. 26 - Prob. 92IAECh. 26 - Prob. 93IAECh. 26 - Prob. 94IAECh. 26 - Prob. 95IAECh. 26 - For each of the following molecules (a) draw the...Ch. 26 - Prob. 97FPCh. 26 - Prob. 98SAECh. 26 - Explain the important distinctions between each...Ch. 26 - Describe the characteristics of each of the...Ch. 26 - The compound isoheptane is best represented by the...Ch. 26 - Prob. 102SAECh. 26 - Prob. 103SAECh. 26 - Prob. 104SAECh. 26 - Assign configurations, R or S, to the chiral...Ch. 26 - Consider the following pairs of structures In each...Ch. 26 - Prob. 107SAECh. 26 - Prob. 108SAECh. 26 - Prob. 109SAECh. 26 - Prob. 110SAECh. 26 - Prob. 111SAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How many isopropene units in this structure?arrow_forward2. Which of the following compounds are optically active? Identify the chiral carbon with an asterisk (*). a. Butan-2-ol b. 2-aminopropanoic acid c. 2,3-dichlorobutane d. 1-phenylethanolarrow_forwardClassify the following monosaccharide. a. deoxy carboxylic acid pentose b. carboxylic acid pentose c. deoxyhexose d. ketopentose e. aldopentosearrow_forward
- Question 4arrow_forwardPart 1 1 Indicate the configuration (R or S) around the chiral carbon(s) in the following compound. Attachments A. SR B. S ⒸC.R OD. SS E. RR F. Molecule is Achiralarrow_forwardClassify the following disaccharide. a. beta 1-2 disaccharide b. alpha 1-4 disaccharide c. alpha 1-6 disaccharide d. alpha 1-2 disaccharide e. beta 1-4 disaccharide f. beta 1-6 disaccharidearrow_forward
- Naming Organic structures: Only use naming systems specially covered in this course, do not use common names 1. Assign a name to each of the following monosaccharides. Use D and L designations. CH₂OH | C=0 a. b. C. a. H- -OH -OH CH₂OH d. True or False: -H b. H-C-OH HO-C-H T CH,OH Circle all chiral centers for the monosaccharides shown. The molecules shown can rotate plane polarized light?arrow_forward5. Explain why ethanol can be oxidized but 2-methyl-2-butanol cannot be oxidized?arrow_forwardHow many chiral carbons are in the sugar below? a. 2 b. 3 c. 5 d. 6 e. 4arrow_forward
- 2. Identify the type of glycosidic bond that links the monosaccharides and the structure of the disaccharide as a- or 8-isomer. CH,OH H. H OH H CH: H но OH H OH H H. OH H. OHarrow_forwardIdentify which type of isomerism is described by each statement. A. Isomerism based on a single difference on the location of OH B. Isomerism based on functional group of the monosaccharides C. Isomerism based on configuration of hemiketal or hemiacetal carbon D. Isomerism based on ability to rotate plane of polarized light E. Isomerism based on OH position in the carbon atoms of the linear structure F. Isomerism based on aromatic ring structure derivationarrow_forwardQuestion 9 Structure A Br H3C...C H CH₂CH3 Structure B CH 3 H..MC Br CH₂CH3 Determine the relationship between the structures above. Are they the same molecule, constitutional isomers, or stereoisomers? If A and B are stereoisomers, futher classify them as diastereomers or enantiomers. A and B are the same molecule A and B are constitutional isomers A and B are stereoisomers and diastereomers A and B are stereoisomers and enantiomersarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY