
Concept explainers
(a)
Interpretation:
To determine whether there is any chiral carbon atom present in the compound.
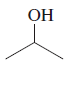
Concept introduction:
Chiral carbon atoms are the species in which a carbon is attached to all different groups.
Since, the valency of a carbon atom is four. So, every chiral carbon atom will be attached to four different groups. A chiral atom is non-superimposable on its mirror image and it is unsymmetrical in nature.
Whereas a carbon atom that is superimposable on its mirror image is called an achiral carbon atom. It is symmetrical in nature.
(b)
Interpretation:
To determine whether there is any chiral carbon atom present in the compound.
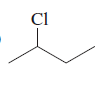
Concept introduction:
Chiral carbon atoms are the species in which a carbon is attached to all different groups.
Since, the valency of a carbon atom is four. So, every chiral carbon atom will be attached to four different groups. A chiral atom is non-superimposable on its mirror image and it is unsymmetrical in nature.
Whereas a carbon atom that is superimposable on its mirror image is called an achiral carbon atom. It is symmetrical in nature.
(c)
Interpretation:
To determine whether there is any chiral carbon atom present in the compound.
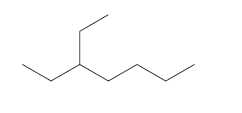
Concept introduction:
Chiral carbon atoms are the species in which a carbon is attached to all different groups.
Since, the valency of a carbon atom is four. So, every chiral carbon atom will be attached to four different groups. A chiral atom is non-superimposable on its mirror image and it is unsymmetrical in nature.
Whereas a carbon atom that is superimposable on its mirror image is called an achiral carbon atom. It is symmetrical in nature.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EBK GENERAL CHEMISTRY
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardWhat is meant by the term “unsaturated hydrocarbon”? What structural feature characterizes unsaturated hydrocarbons?arrow_forwardDrawthe characteristic functional group of FOUR of the following six families of organic compounds: Alcohol, Amine, Aldehyde, Ketone, Carboxylic acid, or Ester.arrow_forward
- The following structural diagrams represent organic compounds with common industrial uses. Match the structural diagrams above with their classifications given below. Note: a number can be used more than once. Alcohol =? Aromatic =? Carboxylic acid =? Halogenated hydrocarbon =? (Picture of equation attached)arrow_forwardIdentify the functional group CH3-C 0-CH2-CH2-CH3arrow_forwardWhat is the difference between a structural isomer and structural conformations of an organic molecule?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





