
Concept explainers
Interpretation:
The relationship between the boxed atom and the circled atoms in the given structure has to be given.
Concept Introduction:
Cis, Trans Isomerism in Bicycloalkanes:
Depending on the orientation of two hydrogen atoms at the ring junction, whether the hydrogens are cis or trans to each other, two stereoisomers are possible for bicycloalkanes.
Example: Decalin:
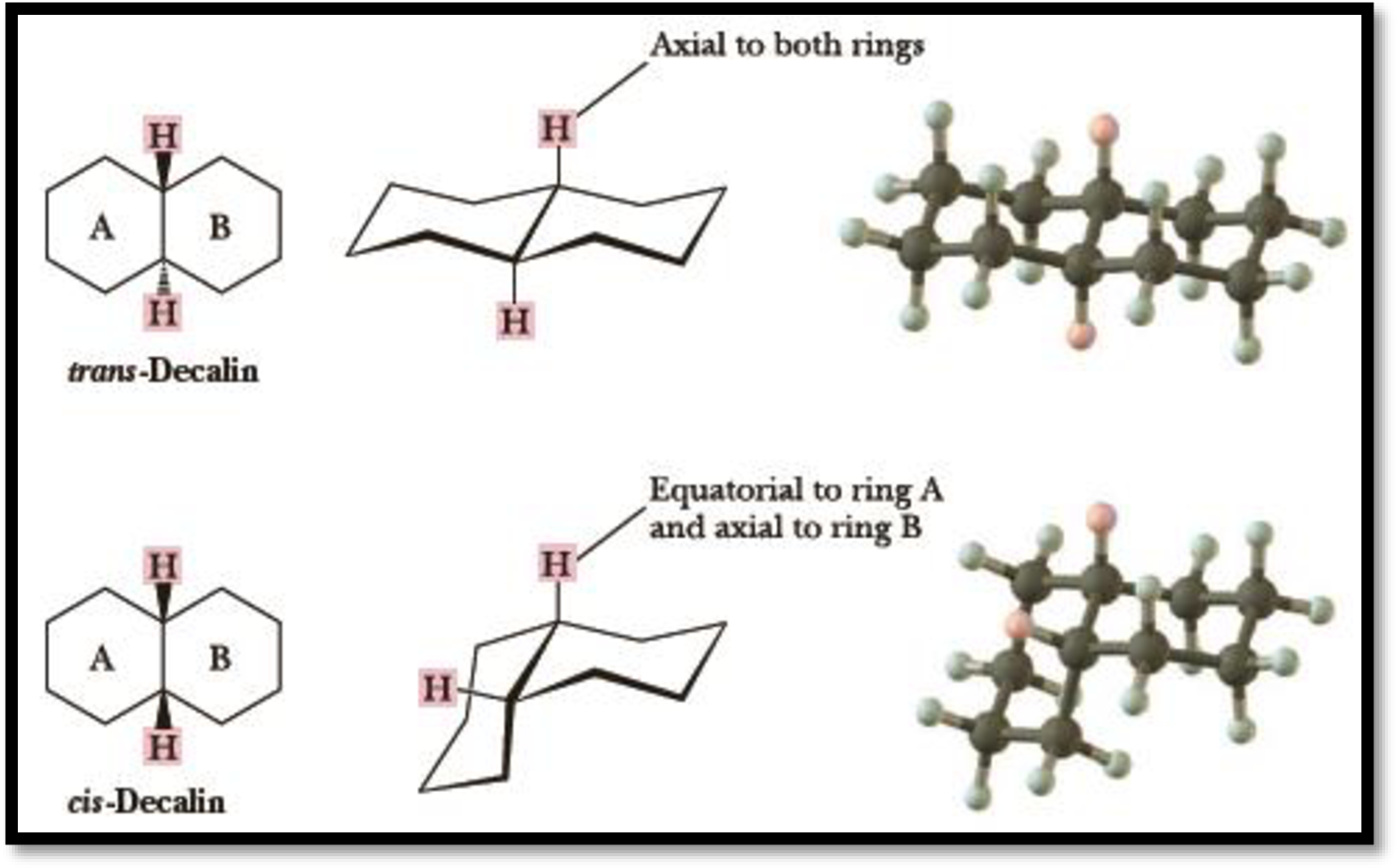
Figure 1
Since both the ring in bicycloalkane is locked into one chair conformation, neither ring can invert to its alternative chair and thus the other substituent on ring which would be in equatorial position will remian equatorial and the substituent which would be in axial position will remain axial.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Chloramlhenicol, a powerful antibiotic in 1947 from the streptomyces venezuelae bacterium, is active against a broad spectrum of bacterial infections and is particularly valuable against typhoid fever. Assign R or S configurations to the chirality centers in chloramphenicolarrow_forwardTamiflu(shown below) is an antiviral medication used to treat influenza. How many asymmetric centers are present in the this structure?arrow_forwardExplain the result of the specific rotation (-130\deg) shown by that compound (C13H1002) based on its molecular structure visualized in three dimensions.arrow_forward
- Which molecule would have their 2 chair conformations be of equal energies?arrow_forwardChloramphenicol is a broad-spectrum antibiotic that is particularly useful against typhoid fever. What is the configuration of each of its asymmetric centers?arrow_forward5. For the molecules below assign the stereochemical configuration of the selected tetrahedral carbon chiral centers (R, S or N (not a chiral center)) and the alkene (E, Z or N (not a stereocenter)) that are indicated by the arrows (note that you do not have to assign the configuration of every chiral center in the molecule). If the atom in question is not a chiral center or is not a stereocenter circle N for neither. b) RSN RSN HO" RSN EZ N RSN НО. H₂N HO HO OH HO OH RSN RSN CH3 НИ H3 CH₂ RSN RSN RSN EZ N RSNarrow_forward
- The diaxial conformation of cis-1, 3-dimethylcyclohexane is approximately 23 kJ/mol (5.4 kcal/mol) less stable than the diequatorial conformation. Draw the two possible chair conformations, and suggest a reason for the large energy difference.arrow_forwardcis-1,2-Dimethylcyclohexane is optically inactive even though it has two chirality centers. Explain.arrow_forwardmyo-Inositol, one of the isomers of 1,2,3,4,5,6-hexahydroxycyclohexane, acts as a growth factor in both animals and microorganisms. Draw the most stable chair conformation of myo-inositol.arrow_forward
- Wat are the four alcohols with the molecular formula C4H9OH. How many of these are chiral?arrow_forwardCompare the two possible chair conformations of this molecule. Given the AG for the two possible chair conformations, calculate the equilibrium constant for the interconversion between the two chair conformers at room temperature. CI H3C AG = 1.3 kcal/mol %3D AG = -RT In K %3D rearranges to K = e-AG/RT %3D R = 1.987 x 10-3 kcal/molK ||arrow_forwardIs cis-1,2-dibromocyclohexane a meso compound? In its planar structure, it has a line of symmetry but it has two chiral carbons. However, in its chair conformation, it does not look like it has a line of symmetry but it has two chiral carbons.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



