
Concept explainers
Describe what is meant by each of the following reaction types, and illustrate with an example:
(a) nucleophilic substitution reaction: (b) electrophilic substitution reaction; (c) addition reaction;
(d) elimination reaction, (e) rearrangement reaction.
(a)
Interpretation:
The nucleophilic substitution reaction should be defined with example.
Concept introduction:
Nucleophilic substitution reaction describes the attack of the electron-rich group that is nucleophile on electron deficient groups that is electrophile.
Answer to Problem 1E
Nucleophilic substitution reaction is the type of reaction in which the nucleophile (electron rich species) attacks the electron-deficient carbon atom which is electrophilic.
Explanation of Solution
Nucleophilic substitution reaction is defined as an organic reaction which includes the attack of a nucleophile on electrophilic center along with the removal of the leaving group.
The example of the nucleophilic substitution reaction is,
In this reaction, chlorine of chloroethane is replaced by a hydroxyl group
(b)
Interpretation:
The electrophilic substitution reaction should be defined with example.
Concept introduction:
The electrophilic substitution reaction describes the displacement of functional group or hydrogen atom by an electron deficient group or electrophile.
Answer to Problem 1E
Electrophilic substitution reaction is defined as the organic reaction in which the electrophile replaces a functional group of a compound or hydrogen atom.
Explanation of Solution
Electrophilic substitution reaction is defined as the organic reaction which includes the replacement of functional group or
Example of electrophilic substitution reaction is,
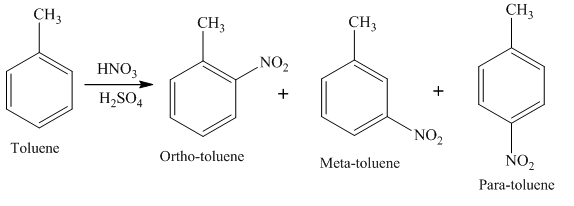
In this reaction, Toluene undergoes electrophilic substitution to form para nitrotoluene, meta nitrotoluene, and ortho nitrotoluene.
(c)
Interpretation:
The addition reaction should be defined with example.
Concept introduction:
The addition reaction describes the combination of two or more smaller molecules to form a larger molecule.
Answer to Problem 1E
Addition reaction is defined as the reaction in which two or more molecules combine to form a single and large molecule.
Explanation of Solution
The reaction of the addition of the two or more reactants that is A and B to produce a single product C is termed as addition reaction.
The example of addition reaction is,
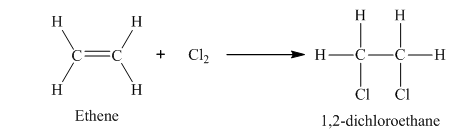
In this reaction, chlorine molecule combines with ethene to form 1, 2-dichloroethane.
(d)
Interpretation:
The elimination reaction should be defined with example.
Concept introduction:
The elimination reaction describes the removal of two substituents from the reactant molecule to form the product.
Answer to Problem 1E
Elimination reaction is the reaction by which the reactant molecule or compound breaks into two or more products.
Explanation of Solution
Elimination reaction is the type of reaction in which two substituents are removed from the reactant molecule to form the product. Generally, unsaturated compounds are formed in an elimination reaction.
The example of the elimination reaction is,
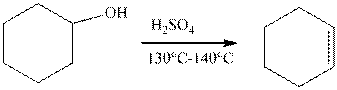
The reaction of cyclohexanol in the presence of
(e)
Interpretation:
The rearrangement reaction should be defined with example.
Concept introduction:
The rearrangement reaction describes the rearrangement of bonds in a molecule to form the product.
Answer to Problem 1E
It is the process of movement of bonds within a molecule to give rise to structural isomers.
Explanation of Solution
It is defined as a reaction in which an atom or a bond migrates from one atom in reactant molecule to adjacent atom to give rise to the product.
Example of rearrangement reaction is,
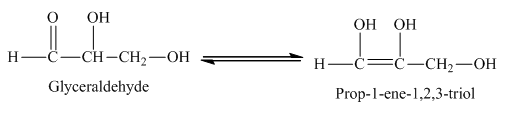
The glyceraldehyde undergoes rearrangement to form enediol.
Want to see more full solutions like this?
Chapter 27 Solutions
Mastering Chemistry With Pearson Etext -- Standalone Access Card -- For General Chemistry: Principles And Modern Applications (11th Edition)
Additional Science Textbook Solutions
Organic Chemistry (9th Edition)
Chemistry In Context
Chemistry For Changing Times (14th Edition)
EBK INTRODUCTION TO CHEMISTRY
Elementary Principles of Chemical Processes, Binder Ready Version
- Both alcohols and carboxylic acids undergo substitution,but the processes are very different. Explainarrow_forwardIs the reaction between an alkene and a halogen a substitution reaction or some other reaction? Explain the reaction mechanism by giving an example.arrow_forwardC12H17O2N ----> CH3I gives ( in structural formula ) ?arrow_forward
- Is the reaction between an alkene and a halogen a substitution reaction(displacement reaction) or another reaction? Explain the reaction mechanism by giving an example.arrow_forwardGive the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardEncricle the functional groups in the followingf compounds, label each with the letters indicated and indentify the class in which the functional group may be foundarrow_forward
- Explain why benzene can not undergo electrophilic addition reaction, but can only do electrophilic substitution reaction?arrow_forwarda. Explain the differences between a pendant and stiffening groups in polymers.b. What is the effect of stiffening and softening groups on the properties ofpolymers? Explain by using suitable examples.arrow_forward(a) Differentiate between copolymerization and homopolymerization. Give one example of each.(b) What is the role of Benzoyl peroxide in preparation of Polythene?arrow_forward
- (a) One test for the presence of an alkene is to add a smallamount of bromine, which is a red-brown liquid, and lookfor the disappearance of the red-brown color. This test doesnot work for detecting the presence of an aromatic hydrocarbon.Explain. (b) Write a series of reactions leading topara-bromoethylbenzene, beginning with benzene andusing other reagents as needed. What isomeric side productsmight also be formed?arrow_forwardWhich of these three compounds Alkane , Alkene and Aromatics has an exothermic reaction with concentrated sulfuric acid? Why?arrow_forwardHow can alkenes and alkynes be classified: as nucleophiles, as electrophiles or depending on the reaction conditions could be both?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


