
The reason that the electrons in the Rutherford model fall into the nucleus.
Answer to Problem 1RQ
Solution:
The reason for electrons to fall into the nucleus, according to the Rutherford atomic model, is the force of attraction between the positive nucleus and the negatively-charged electron.
Explanation of Solution
Introduction:
The Rutherford atomic model is also known as the planetary or nuclear model of an atom. This model designated the atom as a positively charged, dense, tiny core called as a nucleus. Electrons (negative constitutent of atom) constitute the mass of the atom. These electrons revolve at a certain distance from the nucleus similar to the way planets revolve around the Sun.
Explanation:
According to Rutherford’s postulate, electrons revolve at a very high speed around the nucleus of an atom in a particular orbit. However, accelerated charged particles release a certain amount of
The electron spirals inward towards the nucleus as shown in the diagram:
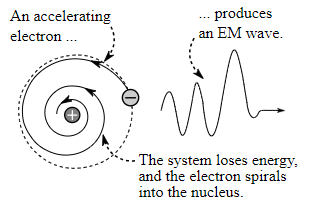
The given figure shows the emission of electromagnetic radiation by the accelerating electron as well as the shrinkage of the orbit of the electron.
Conclusion:
The force of attraction between the positive nucleus and the negatively-charged electron is the main cause for electrons to fall into the nucleus, according to the Rutherford atomic model.
Want to see more full solutions like this?
Chapter 28 Solutions
College Physics
- What two pieces of evidence allowed the first calculation of me, the mass of the electron? (a) The ratios qe/me and qp/mp. (b) The values of qe and EB. (c) The ratio qe/me and qe. Justify your response.arrow_forward. Figure 10.47 is the energy-level diagram for a particularly simple, fictitious element, Vernium (Vn). Indicate by the use of arrows all allowed transitions leading to the emission of photons from this atom and order the frequencies of these photons from highest (largest) to lowest (smallest).arrow_forwardWhy are UV, x rays, and rays called ionizing radiation?arrow_forward
- Integrated Concepts Particles called muons exist in cosmic rays and can be created in particle accelerators. Muons are very similar to electrons, having the same charge and spin, but they have a mass 207 times greater. When muons are captured by an atom, they orbit just like an electron but with a smaller radius, since the mass in aB=h242mekqe2=0.5291010m is 207 me. (a)Calculate the radius of the n=1 orbit for a muon in a uranium ion (Z=92). (b) Compare this with the 7.5-fm radius of a uranium nucleus. Note that since the muon orbits inside the electron, it falls into a hydrogen-like orbit. Since your answer is less than the radius of the nucleus, you can see that the photons emitted as the muon falls into its lowest orbit can give information about the nucleus.arrow_forwardWhat are isotopes? Why do different isotopes of the same element have similar chemistries?arrow_forwardRutherford fired a beam of alpha particles (helium nuclei) at a thin sheet of gold. An alpha particle was observed to be deflected by 90.0; its speed was unchanged. The alpha particles used in the experiment had an initial speed of 2 107 m/s and a mass of 6.7 1027 kg. Assume the alpha particle collided with a gold nucleus that was initially at rest. Find the speed of the nucleus after the collision.arrow_forward
- In Millikan's oil-drop experiment, one looks at a small oil drop held motionless between two plates. Take the voltage between the plates to be 2033 V, and the plate separation to be 2.00 cm. The oil drop (of density 0.81 g/cm3) has a diameter of 4.0106 m. Find the charge on the drop, in terms of electron units.arrow_forwardIntegrated Concepts A 1.00-fm photon has a wavelength short enough to detect some information about nuclei. (a) What is the photon momentum? (b) What is its energy in joules and MeV? (c) What is the (relativistic) velocity of an electron with the same momentum? (d) Calculate the electron's kinetic energy.arrow_forwardDefine the quantum numbers n,l,ml,s, and ms.arrow_forward
 Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





