
(a)
Interpretation: The mode of ring closure for each of the given electro cyclic reactions has to be identified.
Concept introduction:
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”
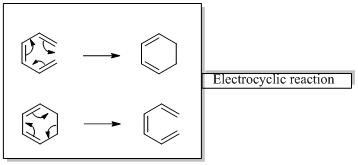
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
In conrotatory mode both the atomic orbitals of the end groups turn in the same direction.
But in the disrotatory the turning direction is different.
(b)
Interpretation: The configuration of the indicated hydrogens in the reactions has to be predicted.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
ORGANIC CHEMISTRY 345 WSU >IP<
- The molecules below react in a displacement reaction. Determine the products) and assign configuration (R/S) to any stereogenic centers in the product. The Cahn-Ingold - Prelog rules can be used to assign priority to the four different groups on a stereogenic center.arrow_forwarda. Identify the mode of ring closure for the following electrocyclic reactions.b. Are the indicated hydrogens cis or trans?arrow_forwardClassify the sigmatropic rearrangement with bracketed numbers.arrow_forward
- Explain the stereoselecticity of this reactionarrow_forwardUsing FMO theory, determine whether a carbanion rearrangement, analogous to the 1,2-hydride shift in Equation D-10, is allowed or forbidden.arrow_forwardDraw the products for these Diels-Alder reactions with correct relative (or absolute) configuration of all formed stereocenters. Explain the stereochemical outcome assuming the involvement of secondary interactions whenever possible.arrow_forward
- This is a Diels-Alder reaction between cyclopentadiene and maleic anhydride (endo and exo products). For each cycloaddition product, draw in all hydrogen atoms, and write the molecular formula below.arrow_forwardIn some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer ispresent. For example, treatment of optically pure 1-bromo-1phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.arrow_forwardExplain how substituents on thearomatic ring promote substitutionat some positions but not at othersarrow_forward
- What is the product E and how is it formed? Can this be explained using frontier molecular orbital analysis? What would the regiochemistry and stereochemistry be?arrow_forwardA)Circle all of the stereo centers in MDMA. B) assign the absolute stereochemistry (R or S) for each stereo centerarrow_forwardIn some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

