
Concept explainers
Interpretation:
The differences in the products obtained under photochemical and thermal condition should be accounted for the given reaction.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In a sigmatropic rearrangement reaction “ one new sigma-bond is formed as another breaks.”
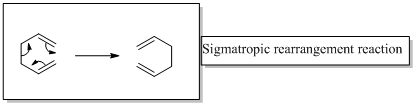
Sigmatropic rearrangement reactions are designated with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Woodward – Hoffmann rules for sigmatropic rearrangement reactions are listed below
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
ETEXT+MASTERINGCHEMISTRY STANDALONE AC
- A) Provide the reagent and reaction mechanism to show how the reactants and products in the following reaction can interconvert B) under what conditions would the reaction I) favour reactants, II) favour the products and III) why?arrow_forwardDefine Thermal Electrocyclic Reactions ?arrow_forwardThe aerobic oxidation of para-xylene to terephthalic acid is an important process in industrial chemistry. Discuss why the oxidation of the second methyl group requires harsher conditions than the oxidation of the first methyl group. You should accurately reference all your bibliographic material.arrow_forward
- Propose a single synthesis method for cobalt(II) sulphate (CoSO4.7H2O) and describe in detail.arrow_forwardBriefly outline the Woodward-Hoffman description and explain how they can be used to predict if a pericyclic reaction will occur thermally or photochemically.arrow_forwardOutline the possible reaction pathways with proper mechanism for the formation ofNylon 6 from Benzene molecule.arrow_forward
- Define the mechanism of acid–catalyzed halogenation ?arrow_forwardDescribe the species that would result from the subsequent transfer of the proton from the metal to one of the Cp rings of ferrocene. Give the formal oxidation state of the metal centre and the valence electron countarrow_forwardSuggest the mechanisms usedarrow_forward
- The reaction between peroxodisulphate (VI) ions, S2O82- and iodide ions, I- can be catalyzed by iron(III) ions, Fe3+. Suggest a mechanism for the catalytic reaction.arrow_forwardFor a diene halogenation reaction at 0 degrees Celsius, assume that 2.50 g of 1,3-butadiene is used, 50 mL of ether is used as the solvent, and the concentration of HCl is 3.00M. What is the volume of HCl required to preform a diene halogenation reaction and the theoretical yield of products of the reaction. (Please include the number of moles of each reactant and product)arrow_forwardDetermine all of the products obtained from the addition of HCl to the 1,3-diene. Once determined, draw a mechanism that accounts for the formation of every product. Then, Identify and account for the formation of the major adduct/or adducts under these conditions assuming that the reaction is conducted under thermodynamic conditions.arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
