
Concept explainers
Interpretation:
It should be determined that the given [1, 7] sigmatropic rearrangement involves whether suprafacial or antarafacial rearrangement.
Concept introduction:
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
When hydrogen migrates in a sigmatropic rearrangement, the s orbital of the hydrogen is partially bonded to both the migration origin and the migration terminus in the transition state.
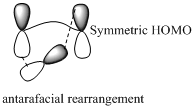
Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Migration of carbon occurs through two ways because it has a two lobed p orbital. Carbon can simultaneously interact with the migration origin and the migration terminus using one lobe of its p orbital. Migration of carbon in suprafacial and antarafacial rearrangement can be represented as follows,
Carbon migrating with one lobe of its p orbital interacting


Carbon migrating with both lobe of its p orbital interacting

Trending nowThis is a popular solution!

Chapter 28 Solutions
Pearson eText Organic Chemistry -- Instant Access (Pearson+)
- 9 A disaccharide consists of the molecules of D-glucose and D-glucosamine joined by a (1a, 1b)-glucosidic bond. a) Draw the structure of the disaccharide (Haworth projection) if you know that in D- glucose the anomeric carbon has an a-configuration. b) Give the name of the union you drew in question a. c) Draw the structure of the product (cantilever conformation) resulting from the effect of excess acetic anhydride and pyridine on the disaccharide. Hint: D-glucosamine has the same structure as D-glucose, but C-2 has an amino group instead of a hydroxyl.arrow_forwardWhat is the maximum number of stereoisomers possible for a compound with: (a) three stereogenic centers; (b) eight stereogenic centers?arrow_forwardDraw and name all the stereoisomers of 2-bromo-3-iodopentane. Indicate which stereoisomers are enantiomers and which are diastereomers.arrow_forward
- 34 (a) Find all of the carbon stereocenters in a choles- terol molecule. oled ti 290b (b) How many total stereoisomers are possible? (c) How many of these stereoisomers do you think are found in nature? 54 55arrow_forward8. Identify the stereochemical relationship between the structural pairs shown below as (A) identical structures that are not meso; (B) identical structures that ARE meso; (C) structures that are enantiomers; or (D) structures that are diastereomers. a) b) H3C c) CH3 Br H. CH3 Br Br CH3 H. CI Holl H Cill Cl- CH3 Br H. CI CH3 HO. H. H3C Cl H3C- H- H. CH3 Cl a) b) c) 9. Identify the stereochemical relationship between the structural pairs shown below as (A) identical structures that are not meso; (B) identical structures that ARE meso; (C) structures that are enantiomers; or (D) structures that are diastereomers. a) b) c) CH2CH3 NH2 HO CH3 H3C. CH3 Cl HO H;C CH2CH3 F. F H;C. CI H H,N -CH3 CH3 NH2 H3C Br H2N C1 H3C CH3 CH3 H;CH,C- Cl F H. H Br CH,CH3 CH3 a) b) c)arrow_forwardIn the molecules below (a) H (b) O H-C-OH HO-C-H H-C-OH HO-C-H HO-C-H HO-C-H H-C-OH -CIO CH,OPo, CH,OPO, CH,OPO, CHOPO, Which molecule is the enantiomer of molecule a? which molecule is a diasteromer of molecule b?arrow_forward
- (a) Label compounds A, B, and C as D- or L-sugars. (b) How are compounds A and B related? A and C? B and C? Choose from enantiomers, diastereomers, or constitutional isomers.arrow_forwardIdentify pairs of molecules that represent enantiomers and diastereomers and identify each center stereogenic by writing R or S next to it.arrow_forward(9) How many chiral centers are present in a molecule of 1,3-dimethylcyclohexane? (C) 2 (D) 3 1 (A) O (10) What is the percent composition of a mixture of (S)-(+)-2-butanol, [a] = +13.52°, and (R)-(-)-2-butanol, [a] = -13.52°, with a specific rotation [a]D = -6.76° (A) 75%R 25%S (B) 25%R 75%S (C) 50%R 50%S 67%R 33%Sarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


