
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 29, Problem 29.17P
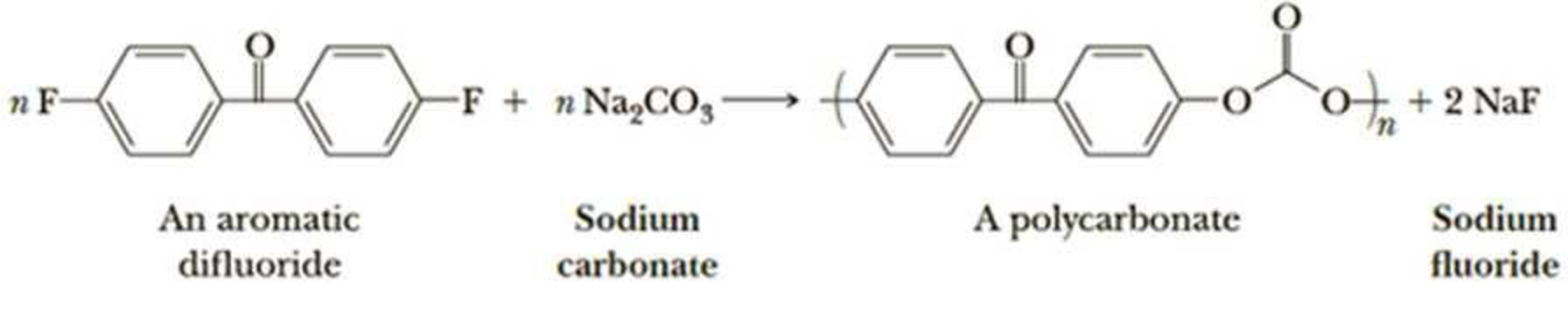
Polycarbonates (Section 29.5C) are also formed by using a nucleophilic
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw (by hand) the monomers used to synthesize poly(ethyl cyanoacrylate) and poly(octyl cyanoacrylate), and chemical structure of the repeat units of each when polymerized.
One common type of cation exchange resin is prepared by polymerization of a mixture containing styrene and 1,4-divinylbenzene . The polymer is then treated with concentrated sulfuric acid to sulfonate a majority of the aromatic rings in the polymer.
Q.) Explain how this sulfonated polymer can act as a cation exchange resin
Kodel is a condensation polymer made from terephthalic acid and 1,4-cyclohexanedimethanol. Write the structure of the resulting polymer.
Chapter 29 Solutions
Organic Chemistry
Ch. 29.2 - Prob. 29.1PCh. 29.5 - Prob. 29.2PCh. 29.6 - Prob. AQCh. 29.6 - Prob. BQCh. 29.6 - Prob. CQCh. 29.6 - Prob. DQCh. 29.6 - Prob. EQCh. 29.6 - Prob. FQCh. 29.6 - Prob. 29.3PCh. 29.6 - Prob. 29.4P
Ch. 29 - Prob. 29.5PCh. 29 - Prob. 29.6PCh. 29 - Prob. 29.7PCh. 29 - Prob. 29.8PCh. 29 - Prob. 29.9PCh. 29 - Prob. 29.10PCh. 29 - Prob. 29.11PCh. 29 - Prob. 29.12PCh. 29 - Prob. 29.13PCh. 29 - Prob. 29.14PCh. 29 - Prob. 29.15PCh. 29 - Prob. 29.16PCh. 29 - Polycarbonates (Section 29.5C) are also formed by...Ch. 29 - Prob. 29.18PCh. 29 - Prob. 29.19PCh. 29 - Prob. 29.20PCh. 29 - Prob. 29.21PCh. 29 - Draw a structural formula of the polymer resulting...Ch. 29 - Prob. 29.23PCh. 29 - Prob. 29.24PCh. 29 - Prob. 29.25PCh. 29 - Select the monomer in each pair that is more...Ch. 29 - Prob. 29.27PCh. 29 - Prob. 29.28PCh. 29 - Prob. 29.29PCh. 29 - Prob. 29.30PCh. 29 - Prob. 29.31PCh. 29 - Prob. 29.32PCh. 29 - Prob. 29.33PCh. 29 - Radical polymerization of styrene gives a linear...Ch. 29 - Prob. 29.35PCh. 29 - Prob. 29.36PCh. 29 - Prob. 29.37PCh. 29 - Prob. 29.38P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine whether the reactions occur by elimination, nucleophilic substitution, electrophilic addition, or electrophilic substitution.arrow_forwardA polyester can be formed from the reaction of which compounds? A. Alkenes B. Dialcohols and diammines C. Alkynes D. Dicarboxylic acids and diammes E. Dicarboxylic acids and dialcoholsarrow_forwardDraw the structure of the following polymers and indicate the polymerization type responsible for the givenpolymers.Indicate, where appropriate, the polyester, polyamide or polyurethane linkages.a) Poly(ethene terephthalate)b) Poly(vinyl alcohol)c) Polyisoprene and isomer structures of polyisoprened) Poly(methyl methacrylate)e) Polycaprolactamarrow_forward
- An organic compound is analysed and found to contain 66.7% carbon, 11.2% hydrogen and 22.1% oxygen by mass. The compound boils at 79.6 C. At 100 C and 0.970atm, the vapour has a density of 2.28g/L. The compound has a carbonyl group and cannot be oxidized to a carboxylic acid. Suggest a structure for the compound.arrow_forwardDraw the structure of the polymer formed by chain-growthpolymerization of each monomerarrow_forwardCompound A is a novel poly(ester amide) copolymer that can be used as a bioabsorbable coating for the controlled release of drugs. A is a copolymer of four monomers, two of which are amino acids or amino acid derivatives. The body’s enzymes recognize the naturally occurring amino acids in the polymer backbone, allowing for controlled enzymatic breakdown of the polymer and steady release of an encapsulated drug. Identify the four monomers used to synthesize A; then use Figure 29.2 to name the two amino acids.Figure 29.2: The 20 naturally occurring amino acidsarrow_forward
- (a) Differentiate between copolymerization and homopolymerization. Give one example of each.(b) What is the role of Benzoyl peroxide in preparation of Polythene?arrow_forwardShow how to synthesize carboxylic acids from oxidation of alcohols and aldehydes,carboxylation of Grignard reagents, oxidative cleavage of alkenes and alkynes,hydrolysis of nitriles, and oxidation of alkylbenzenesarrow_forwardPoly(vinyl alcohol), a hydrophilic polymer used in aqueous adhesives, is made by polymerizing vinyl acetate and then hydrolyzing the ester linkages. Vinyl acetate is an ester. Is poly(vinyl acetate) therefore a polyester? Explain.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY