
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 29.6, Problem BQ
Interpretation Introduction
Interpretation:
Among the given terms, the term that explains reason for addition of water to cyanoacrylate has to be identified.
Concept introduction:
Anionic Polymerizations: It can be initiated by addition of a nucleophile to an activated
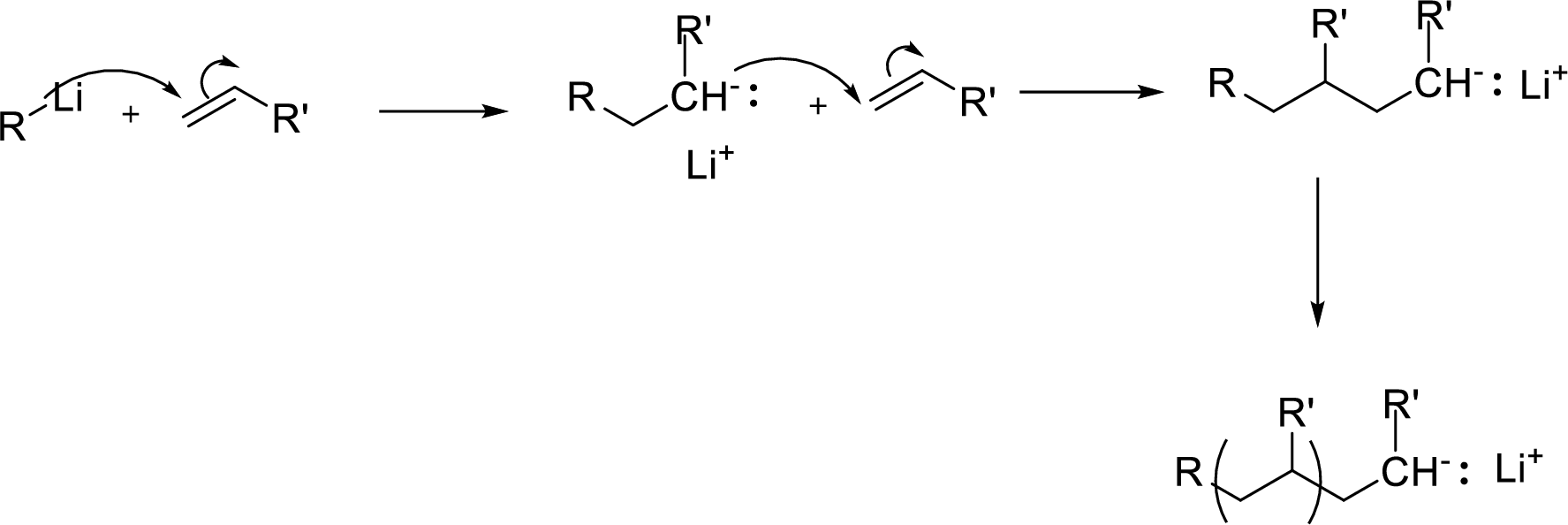
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Why does the weak nucleophile, water, efficiently add to the cyanoacrylate?
1. Superglue must possess hydroxide as a promoter to initiate the polymerization in the presence of water.
2. The carbanion formed by nucleophilic addition of water is resonance-stabilized by two electron-withdrawing groups.
3. The zwitterion formed by addition of water is well solvated in the cyanoacrylate matrix of the glue.
4. Both 1 and 2.
Why do you think initiator molecule is needed to induce the polymerization of ethylene
explain in detail the mechanism for the photoinduced step-growth polymerization of n-ethylcarbazole.
Chapter 29 Solutions
Organic Chemistry
Ch. 29.2 - Prob. 29.1PCh. 29.5 - Prob. 29.2PCh. 29.6 - Prob. AQCh. 29.6 - Prob. BQCh. 29.6 - Prob. CQCh. 29.6 - Prob. DQCh. 29.6 - Prob. EQCh. 29.6 - Prob. FQCh. 29.6 - Prob. 29.3PCh. 29.6 - Prob. 29.4P
Ch. 29 - Prob. 29.5PCh. 29 - Prob. 29.6PCh. 29 - Prob. 29.7PCh. 29 - Prob. 29.8PCh. 29 - Prob. 29.9PCh. 29 - Prob. 29.10PCh. 29 - Prob. 29.11PCh. 29 - Prob. 29.12PCh. 29 - Prob. 29.13PCh. 29 - Prob. 29.14PCh. 29 - Prob. 29.15PCh. 29 - Prob. 29.16PCh. 29 - Polycarbonates (Section 29.5C) are also formed by...Ch. 29 - Prob. 29.18PCh. 29 - Prob. 29.19PCh. 29 - Prob. 29.20PCh. 29 - Prob. 29.21PCh. 29 - Draw a structural formula of the polymer resulting...Ch. 29 - Prob. 29.23PCh. 29 - Prob. 29.24PCh. 29 - Prob. 29.25PCh. 29 - Select the monomer in each pair that is more...Ch. 29 - Prob. 29.27PCh. 29 - Prob. 29.28PCh. 29 - Prob. 29.29PCh. 29 - Prob. 29.30PCh. 29 - Prob. 29.31PCh. 29 - Prob. 29.32PCh. 29 - Prob. 29.33PCh. 29 - Radical polymerization of styrene gives a linear...Ch. 29 - Prob. 29.35PCh. 29 - Prob. 29.36PCh. 29 - Prob. 29.37PCh. 29 - Prob. 29.38P
Knowledge Booster
Similar questions
- The anionic polymerization of ε-caprolactam. Even if the reaction rate between caprolactam is slow, the reaction with lactamanion and acylactam occurs fast. After drawing the structure of the product of the reactions below, explain the reason through the stability of the product.arrow_forwardRadical polymerization of styrene gives a linear polymer. Radical polymerization of a mixture of styrene and 1,4-divinylbenzene gives a cross-linked network polymer of the type shown in Figure 29.1. Show by drawing structural formulas how incorporation of a few percent of 1,4-divinylbenzene in the polymerization mixture gives a cross-linked polymer.arrow_forwardShow the intermediate that would result if the growing chain added to the other end of the styrene double bond. Explain why the final polymer has phenyl groups substituted on every other carbon atom rather than randomly distributedarrow_forward
- Saponified polyvinyl acetate can be reacted with butanal to produce an impact-resistant polymer. What polymer is formed? Name it correctly in the nomenclature and generally assess the possible density of functionalization in such special polymer-analogous modifications.arrow_forwardDraw a chemical reaction scheme for the benzoyl peroxide-initiated polymerization of vinyl chloride showing the initiation, propagation and termination steps. The termination steps should include combination, disproportionation and transfer to initiator. (Assume that there is no secondary decomposition of primary radicals and that regioselective in the propagating step is 100% H-T).arrow_forward(give clear handwritten answer please) Saponified polyvinyl acetate can be reacted with butanal to produce an impact-resistant polymer. What polymer is formed? Name it correctly in the nomenclature and generally assess the possible density of functionalization in such special polymer-analogous modifications!arrow_forward
- Give a mechanism, showing chain branching during the free-radical polymerization of styrene. There are two types of aliphatic hydrogens in the polystyrene chain. Which type is more likely to be abstracted?arrow_forwardA sample of polystyrene is composed of a series of fractions of different sized molecules. a)Calculate the number average and weight average molecular weights of this sample as well as the PDI. Comment on the PDI – is it polydispersed? b)How would adding styrene oligomer change the average molecular weights? Added amount is 5 wt. % of polymer mass and M = 1,000 g/mol. c)What would be the potential polymerisation pathways you may follow to polymerise this polymer d)What would happen if you fractionate the polymer to remove all fractions above 20,000 Da and 50% of any fraction above 15,000 Da? Recalculate the Mn, Mw, and PDI for this new system. e)What type of polymeric materials does this distribution of Mw for the new system corresponds to? Would it be likely solid or liquid at room temperature and atmospheric pressure?arrow_forwardIn the presence of H3O+, 2-methylpropene oxide undergoes chain-growth polymerization such that nucleophilic attack occurs at the more substituted end of the epoxide. Draw a stepwise mechanism for this process, and explain this regioselectivity.arrow_forward
- Based on IR spectra of "Free Radical Polymerization of Styrene" experiment, how well the reaction workedarrow_forwardWhen styrene (vinylbenzene) is commercially polymerized, about 1–3% of 1,4-divinylbenzene is often added to thestyrene. The incorporation of some divinylbenzene gives a polymer with more strength and better resistance to organicsolvents. Explain how a very small amount of divinylbenzene has a marked effect on the properties of the polymerarrow_forwardDraw a graph of UV spectrum of CBZ, ethyl cellulose and PVAarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning