
Concept explainers
(a)
Interpretation:
For the given
(a)
Explanation of Solution
Given polymer structure,
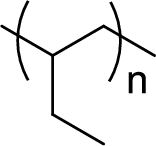
The name of the above polymer is poly(1-butene) because it is formed by the
(b)
Interpretation:
For the given polymer structure, name has to be given.
(b)
Explanation of Solution
Given polymer structure,
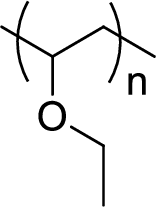
The name of the above polymer is poly(ethyl vinyl ether) because it is formed by the polymerization reaction of ethyl vinyl ether.
(c)
Interpretation:
For the given polymer structure, name has to be given.
(c)
Explanation of Solution
Given polymer structure,
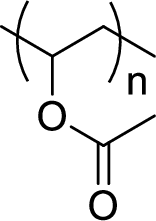
The name of the above polymer is poly(vinyl acetate) because it is formed by the polymerization reaction of vinyl acetate.
(d)
Interpretation:
For the given polymer structure, name has to be given.
(d)
Explanation of Solution
Given polymer structure,
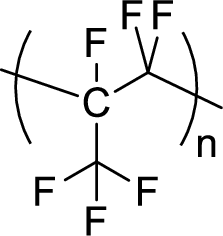
The name of the above polymer is poly(perfluoropropylene) because it is formed by the polymerization reaction of perfluoropropylene.
(e)
Interpretation:
For the given polymer structure, name has to be given.
(e)
Explanation of Solution
Given polymer structure,
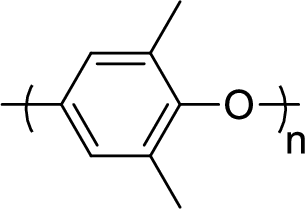
The name of the above polymer is poly(2,6-dimethylphenylene oxide) because it is formed by the polymerization reaction of 2,6-dimethylphenol.
(f)
Interpretation:
For the given polymer structure, name has to be given.
(f)
Explanation of Solution
Given polymer structure,
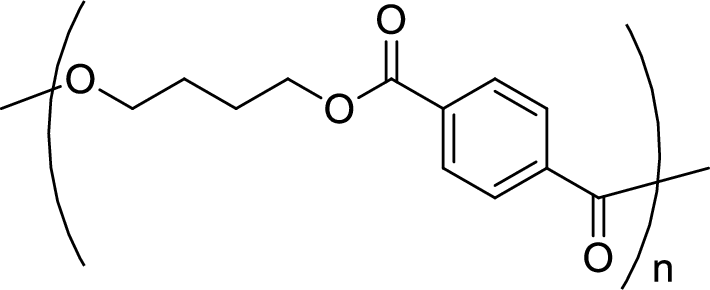
The name of the above polymer is poly(1,4-butylene terephthalate) because it is formed by the polymerization reaction of 1,4-butanediol with terephthalic acid.
(g)
Interpretation:
For the given polymer structure, name has to be given.
(g)
Explanation of Solution
Given polymer structure,
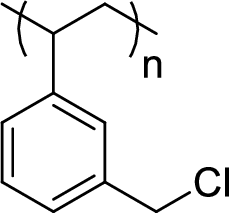
The name of the above polymer is poly(3-chloromethylphenylethylene) because it is formed by the polymerization reaction of 3-chloromethylstyrene.
(h)
Interpretation:
For the given polymer structure, name has to be given.
(h)
Explanation of Solution
Given polymer structure,
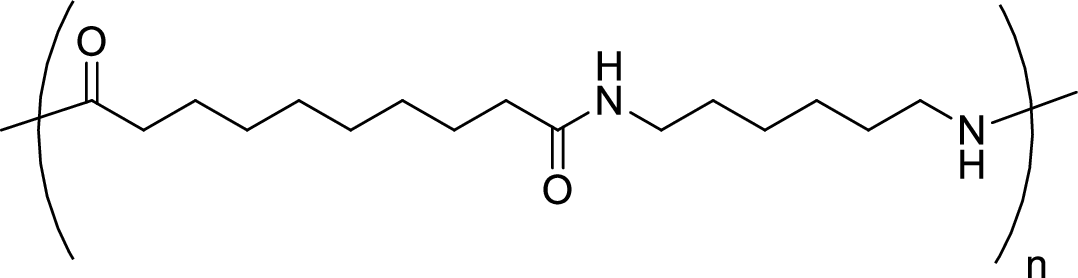
The name of the above polymer is poly(hexamethylene sebacamide) because it is formed by the polymerization reaction of hexamethylenediamine with sebacoyl chloride.
Want to see more full solutions like this?
Chapter 29 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- List TWELVE (12) polymers widely used in high technology application and discuss THREE (3) of the polymers in terms of their production, properties and applications.arrow_forwardName these organic compoundsarrow_forwardCan someone help me name these two organic structures using nomenclature?arrow_forward
- What would be the name of the polymer shown, based on the trend seen with other polymers? An n Polypropylene O Polydipropylene Polyisobutylene O Polybutylenearrow_forwardPolymers are large molecules consisting of two units. Select one: OTrue Falsearrow_forwardsubstances that have the same molecular formula but different structural formulas are known as: a) dimere b) isomers c) polymers d) allotropicarrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





