
Concept explainers
Interpretation:
The slope of the line set between mass and energy released by carbon needs to be determined.
Concept introduction:
The slope of the graph between two components is found by putting one of the components on x-axis and the other on y-axis and then the graph is found with the help of values given.
Answer to Problem 103A
The slope of the graph between mass in gram and the energy released in Kilojoules is a straight line. As the increase in mass also shows the increase in the energy released.
Explanation of Solution
Here is the graph found after setting the values of mass on x-axis and setting the values of energy released on y-axis.
| Mass | Energy released |
| 1 | 33 |
| 2 | 66 |
| 3 | 99 |
| 4 | 132 |
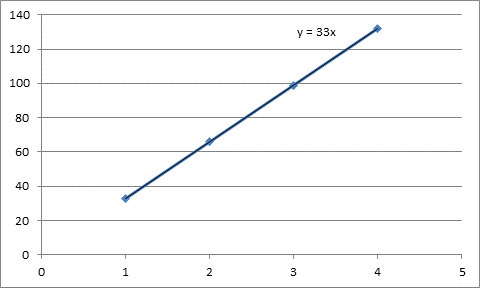
From the straight line on the graph, slope is 33.
The slope of the graph between mass in gram and the energy released in Kilojoules is a straight line. As the increase in mass also shows the increase in the energy released.
Chapter 3 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Introductory Chemistry (5th Edition) (Standalone Book)
Essential Organic Chemistry (3rd Edition)
Chemistry: A Molecular Approach
Organic Chemistry (9th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: Structure and Properties (2nd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





