
Concept explainers
Interpretation:
The curved arrows are to be added to the given reactions, which indicate the flow of electrons for all the bond-forming and bond-breaking steps.
Concept introduction:
Curved arrows are used to represent the direction of the flow of electrons in a reaction mechanism.
Curved arrows are drawn in such a way that they point from the source of an electron pair toward the atom, which receives the electron pair.
The direction of flow of electrons is always from a high electron density site to a low electron density site.
Curved arrows never represent the movement of atoms.
The movement of electrons shown by the curved arrows should not violate the octet rule for the elements, which are present in the second row of the periodic table.
Electrophiles are positive or partially positive species that attract electrons or negatively charged species toward itself.
Answer to Problem 1PP
Solution:
(a)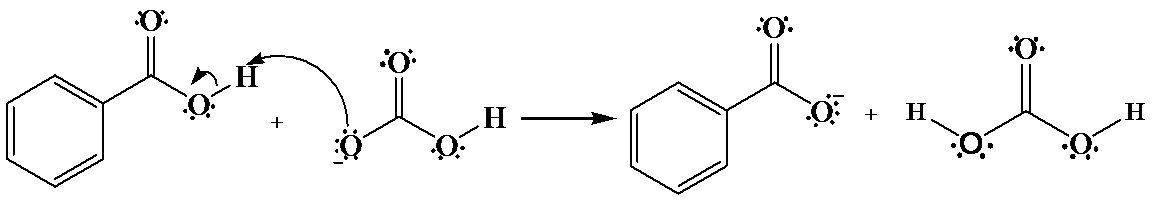
(b) 
Explanation of Solution
a)

The hydrogen of benzoic acid is partially positive and is electrophilic in nature. An oxygen atom provides an unshared pair of electrons, which forms bond with the benzoic acid hydrogen that cause the departure of a benzoate anion.
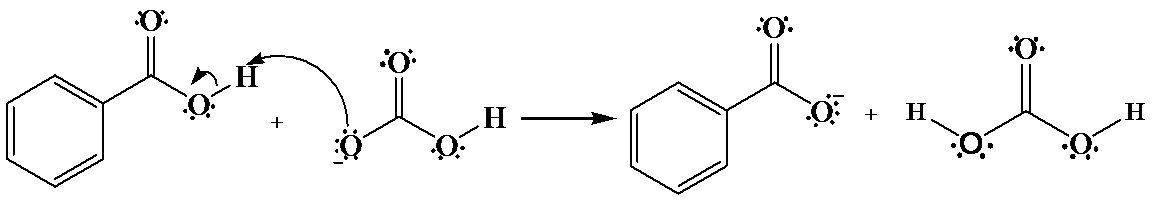
b)
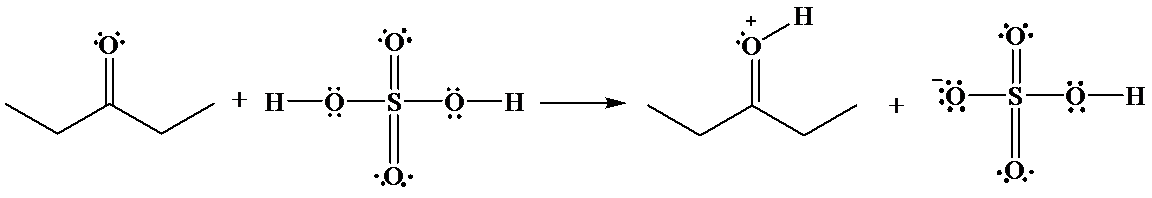
The hydrogen of sulfuric acid is partially positive and is electrophilic in nature. An oxygen atom of ketone provides an unshared pair of electrons, which forms bond with the sulfuric acid hydrogen that cause the departure of a sulphuric acid anion.
The reaction is as follows:

Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
- Please don't provide hand written solution...arrow_forward[References] Draw the expected major organic product of the Sharpless epoxidation of each allylic alcohol using (-)-diethyl tartrate as the chiral catalyst. (a) (b) All hydrogen atoms are implied. If a chiral atom is attached to a hydrogen atom, you should not show the hydrogen atom but use either a wedge or a dashed bond. Apply formal charges where appropriate. Omit lone pairs and radical electrons from your answer. • Omit + signs between structures. ● HO O HO -OCH3 کر ? ChemDoodle Previous Nextarrow_forwardG. (a) Identify the acid and base in each of the following reaction. (b) Show the mechanism by using arrows. (c) For each reaction, what acid-base concept (or definition) is applicable in classifying them? Explain why. Label the acid and base below the structure. Acid-base concept. Explanation Draw arrow (You may copy/paste the reaction in paint or powerpoint to be able to draw the arrow. You may also draw the arrow manually.) 1. :0: II CHIC-H + CH30: :ö: CH3C-H :OCH3 H 2. :0: :0: + H CH3-C-CH3 + CH3-C-CH3 H-arrow_forward
- Please draw a step by step arrow pushing mechanism. Make it very detailed and include all arrows and include ALL resonance structures.arrow_forwardPlease don't provide handwritten solution ...arrow_forwardClassify the following transformation as oxidation, reduction, or neither. Select the single best answer. oxidation reduction neither Give detailed Solution with explanation needed of all options. don't give Handwritten answerarrow_forward
- 6. In an organic chemistry lab, chemists were attempting to convert compound I into compound III via a substitution reaction To their surprise, compound II was the only observed compound after treating compound I with cyanide. "CN observed not observed a. Provide a detailed, stepwise mechaniem for the transformation above that accounts for the formation of product II. Use the curved arrow formalism to show the flow of electrons. Show all lone pairs, intemediates, formal charges, and pertinent resonance structures. b. Explain why product II is formed instead of product III. Use dawings to supportyow answer. C Would you expect compound II to be optically active? Briefly explain. Note The reaction above was run on an enantiomerically pure sample of compound I. CN.arrow_forwardGive detailed Solution with explanation neededarrow_forwardi,ii,iii please.arrow_forward
- Draw the curved arrow mechanism for the reaction between (2R,3S)-3,5-dimethylhexan-2-ol and PCl3. Note the specific instructions for each box. Include nonzero formal charges and lone pairs of electrons on all appropriate atoms. Then answer the question about the mechanism.arrow_forwardI need help please with drawing both of these questionsarrow_forward(a) H. H. 6.44 Which of the following reactions will yield a racemic mixture of products? HCI HBr (a) (c) H2 H2 (b) (d) Pt Ptarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
