
EP PRINCIPLES OF HUM.PHYS.-MOD.MAST.A+P
6th Edition
ISBN: 9780134407166
Author: STANFIELD
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 28E
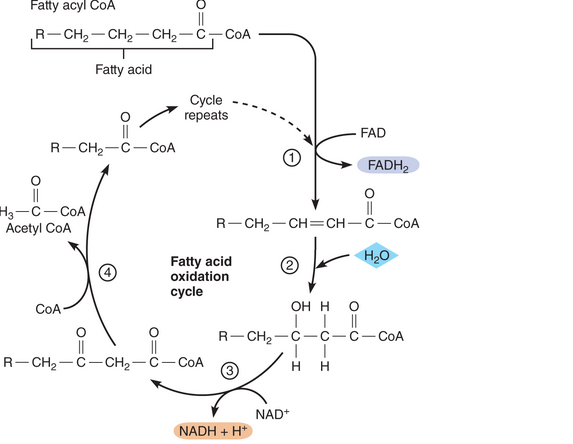
As you know, fatty acids can be oxidized to provide energy for making ATP. This process begins with a set of reactions known as the fatty acid oxidation cycle, or beta oxidation. Before entering the cycle, a fatty acid reacts with coenzyme A to form a molecule called fatty acyl CoA. This molecule then enters the cycle and continues to go through it until all its carbons have been eliminated, as shown on the following page. From your examination of this diagram, answer the following questions for a 1 2-carbon fatty acid.
- How many acetyl CoA molecules will be produced by beta oxidation of this fatty acid? How many ATP molecules can be produced from each acetyl CoA molecule that goes through the Krebs cycle and oxidative phosphorylation?
- How many NAD+ and FAD molecules will be reduced? (Hint: How many bonds must be broken to split a 12-carbon fatty acid into acetyl CoA molecules?)
- How many ATP molecules will be produced per each NADH + H+ and each FADH2 molecule that provides electrons to the electron transport system?
- It takes two ATP molecules to get the cycle started. Based on this information and your answers to the previous questions, how many ATP molecules can be generated from a 1 2-carbon fatty acid?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Saturated fatty acids are degraded by the stepwise reactions of oxidation, producing
acetyl-CoA. Excluding stages 2 and 3 of fatty acid oxidation, how many ATP
molecules would be produced as a consequence of the removal of each Acetyl-CoA
under aerobic conditions?
O O
three
five
two
six
four
The energy for the production of ATP comes from the breakdown of a glucose molecule via
many enzyme-mediated chemical reactions occurring at several distinct stages of cellular
respiration. During this time, the chemical energy stored in glucose is continually trans-
ferred across these reactions.
Which of the following describes the form in which most of the energy from the glucose
molecule is stored just before the oxidative phosphorylation stage in aerobic cellular
respiration?
A
B
с
D
acetyl CoA
FADH 2
NADH
pyruvate
Imagine that you are studying the metabolism of a baby who is feeding on breast milk. You are going to compare the catabolism of two molecules of galactose from lactose in milk with the catabolism of two molecules of leucine from casein in milk. Detail the catabolic path for the molecules until their final oxidation in CO2 + urea.
Chapter 3 Solutions
EP PRINCIPLES OF HUM.PHYS.-MOD.MAST.A+P
Ch. 3.1 - What is the primary distinction between an...Ch. 3.1 - Describe in general terms what occurs in...Ch. 3.2 - In an exergonic reaction, which has more...Ch. 3.2 - Which factor determines the direction of a...Ch. 3.2 - Prob. 3.2.3QCCh. 3.2 - What is activation energy? How does it affect a...Ch. 3.3 - Prob. 3.3.1QCCh. 3.3 - How is the rate of an enzymatic reaction affected...Ch. 3.3 - What is the primary distinction between allosteric...Ch. 3.5 - Prob. 3.4.1QC
Ch. 3.5 - Prob. 3.4.2QCCh. 3.5 - Prob. 3.4.3QCCh. 3.6 - Prob. 1CTQCh. 3.6 - Prob. 2CTQCh. 3.6 - Prob. 3CTQCh. 3.6 - Prob. 3.5.1QCCh. 3.6 - Prob. 3.5.2QCCh. 3.6 - Prob. 3.5.3QCCh. 3.6 - Prob. 3.5.4QCCh. 3.7 - Prob. 3.6.1QCCh. 3.7 - Prob. 3.6.2QCCh. 3.7 - Prob. 3.6.3QCCh. 3 - Prob. 1ECh. 3 - Prob. 2ECh. 3 - Prob. 3ECh. 3 - Prob. 4ECh. 3 - Prob. 5ECh. 3 - Prob. 6ECh. 3 - Prob. 7ECh. 3 - Prob. 8ECh. 3 - Prob. 9ECh. 3 - Prob. 10ECh. 3 - The removal of hydrogen atoms from a molecule is...Ch. 3 - Prob. 12ECh. 3 - Prob. 13ECh. 3 - Prob. 14ECh. 3 - Prob. 15ECh. 3 - Prob. 16ECh. 3 - Prob. 17ECh. 3 - Prob. 18ECh. 3 - Prob. 19ECh. 3 - Prob. 20ECh. 3 - Prob. 21ECh. 3 - Prob. 22ECh. 3 - Prob. 23ECh. 3 - Compare and contrast the mechanisms of...Ch. 3 - Explain how the conversion of pyruvate to lactate...Ch. 3 - Prob. 26ECh. 3 - Prob. 27ECh. 3 - As you know, fatty acids can be oxidized to...Ch. 3 - Prob. 29E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Which carbons of glucose will be incorporated into Palmitic acid by the Fatty Acid Synthase? Multiple answers:Multiple answers are accepted for this question Select one or more answers and submit. For keyboard navigation...SHOW MORE a Carbon 1 b Carbon 2 c Carbon 3 d Carbon 4 e Carbon 5 f Carbon 6 g All carbons will be incorporated. h No carbons will be incorporated.arrow_forwardThe citric acid cycle is shown. The methyl carbon in acetyl CoA is labeled with C14C14 (shown in red). Identify which of the carbons in each intermediate will be labeled in the first round of the cycle by selecting the indicated carbon(s). Each question has multiple options, answering with only one option is incorrect. Which carbon(s) in α‑ketoglutarate will contain C14? 1 2 3 4 5 Which carbon(s) in succinyl‑CoA will contain C14? 1 2 3 4 Which carbon(s) in succinate will contain C14? 1 2 3 4 Which carbon(s) in fumarate will contain C14? 1 2 3 4 Which carbon(s) in malate will contain C14? 1 2 3 4 Which carbon(s) in oxaloacetate will contain C14? 1 2 3 4arrow_forwardTake a look at the citric acid cycle. Look at the entry of acetyl CoA as the first step and oxaloacetate as the final. Determine each absorbed amino acid's probable point of entrance in the cycle and indicate the stoichiometric quantity of the citric acid cycle intermediate or starting material generated per oligopeptide molecule. If an amino acid has many entrance points into the cycle, you may choose any of them as long as stoichiometric connections are maintained.arrow_forward
- When the acetyl-CoA produced during β-oxidation in the liver exceeds the capacity of the citric acid cycle, the excess acetyl-CoA forms ketone bodies—acetone, acetoacetate, and D-b-hydroxybutyrate. This occurs in severe, uncontrolled diabetes: because the tissues cannot use glucose, they oxidize large amounts of fatty acids instead. Although acetyl-CoA is not toxic, the mitochondrion must divert the acetyl-CoA to ketone bodies. What problem would arise if acetyl-CoA were not converted to ketone bodies? How does the diversion to ketone bodies solve the problem?arrow_forwardThe citric acid cycle is shown. The methyl carbon in acetyl CoA is labeled with C14C14 (shown in red). Identify which of the carbons in each intermediate will be labeled in the first round of the cycle by selecting the indicated carbon(s). Each question has multiple options except citrate. Which carbon in citrate will contain C14? 4 1 3 2 Which carbon(s) in isocitrate will contain C14? 1 2 3 4 5 6 Which carbon(s) in α‑ketoglutarate will contain C14? 1 2 3 4 5 Which carbon(s) in succinyl‑CoA will contain C14? 1 2 3 4 Which carbon(s) in succinate will contain C14? 1 2 3 4 Which carbon(s) in fumarate will contain C14? 1 2 3 4 Which carbon(s) in malate will contain C14? 1 2 3 4 Which carbon(s) in oxaloacetate will contain C14? 1 2 3 4arrow_forwardThe conversion of pyruvate to Acetyl-CoA involves an oxidative decarboxylation. Correct the path of the electrons through this mechanism: Pyruvate -> NAD -> lipoamide -> FAD -> TPParrow_forward
- Fourteen NADPH molecules are required to produce one molecules of palmitic acid from acetyl CoA. Substantiate this statement by referring to the enzymatic activities involved in reduction steps during fatty acid synthesis and the number of cycles required to produce palmitic acid from acetyl CoA. How many molecules of ATP is required for the synthesis of palmitic acid from cytosolic acetyl-CoA?arrow_forward3-hydroxy-3-methyl-glutaryl-CoA reductase (HMG-CoA reductase) is a very long name for the enzyme that is involved in the production of cholesterol in the human liver. The enzyme catalyzes a reaction in which electrons are exchanged between reactants to form products. This an example of which of the following chemical reactions that are involved in maintaining homeostasis? HMG-CoA reductase catalyzes a catabolic reaction because electrons are exchanged between the reactants to form products. HMG-CoA reductase catalyzes a specific type of exchange reaction called an oxidation-reduction reaction in which electrons are exchanged between reactants to form products. HMG-CoA reductase catalyzes an anabolic reaction because electrons are exchanged between the reactants to form products. HMG-CoA reductase catalyzes a specific type of oxidation-reduction reaction called an exchange reaction in which atoms are exchanged between reactants to form products. Like most enzymes, HMG-CoA reductase…arrow_forwardTwo carbon atoms are fed into the citric acid cycle as acetyl-CoA. In what form are two carbon atoms removed from the cycle?arrow_forward
- Phosphofructokinase (PFK-1) is one of the most regulated enzymes in metabolism. Based on all of the principles we have learned in this class, which of the following would be most likely to be an allosteric activator of PFK-1? a Pyruvate b Acetyl CoA c Citric acid d glucosearrow_forwardAssume that the following fatty acid is synthesized from acetyl-CoA in the cytosol by fatty acidsynthase. Which of the following statements is true? The last two carbon atoms that are added to the fatty acid are carbon atoms #15 and #16 Synthesis of the fatty acid needs 8 molecules of NADPH + H+ Both A and B Neither A nor Barrow_forwardFor each of the following conditions, state whether this condition would be occurring during gluconeogenesis, glycolysis, or both in liver cells. a. Transporting malate from the cytosol into the mitochondria. _____________ b. Transporting pyruvate from the cytosol into the mitochondria. _____________ c. We would have an increased (or increasing) concentration of F26BP. __________ d. Increased concentration of F6P.__________ e. Hexokinase IV (or glucokinase) is found in the nucleus of the cell. __________ f. The enzyme that synthesizes/breaks down F26BP is phosphorylated. __________ g. Elevated levels of glucagon in the blood. ____________arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Anaerobic Respiration; Author: Bozeman Science;https://www.youtube.com/watch?v=cDC29iBxb3w;License: Standard YouTube License, CC-BY