
To explain:
with a sketch why the frequencies of long-wavelength waves of
Answer to Problem 3.14QA
Solution:
The frequencies of long-wavelength waves of electromagnetic radiations are lower than those of short-wavelength waves.
Explanation of Solution
1) Formula:
i.
ii.
2) Concept:
Frequency, wavelength and energy are related to each other by the above formula.
Let’s take an example of transition of electron from (a) n=3 to n=2 and (b) n= 3 to n=1
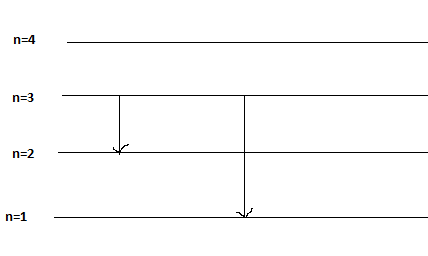
2. Calculations:
From the sketch and calculations, we can say that shorter transition of electron (n=3 to n=2), the wavelength is large thus frequency is short. And for longer transition of electron (n=3 to n=1), the wavelength is small therefore the frequency is large.
Conclusion:
The frequencies of long-wavelength wave of electromagnetic radiations are lower than those of short-wavelength waves.
Want to see more full solutions like this?
Chapter 3 Solutions
Chemistry: An Atoms-Focused Approach (Second Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





