
Principles of Instrumental Analysis
7th Edition
ISBN: 9781337468039
Author: Skoog
Publisher: Cengage
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3, Problem 3.22QAP
Interpretation Introduction
Interpretation:
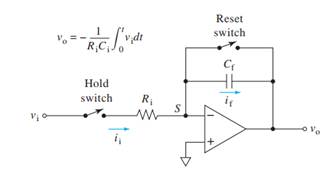
A circuit to perform
Concept introduction:
Operational amplifiers can be used to perform various mathematical operations.
Here shows the addition of the results of two integrations. The diagram shown in following figure illustrates the integration operation using operational amplifier.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The Weibull distribution is widely used in statistical problems relating to aging of solid insulating materials subjected to aging and stress. Use this distribution as a model for time (in hours) to failure of solid insulating specimens subjected to AC voltage. The values of the parameters depend on the voltage and temperature; suppose ? = 2.2 and ? = 220.
(a)
What is the probability that a specimen's lifetime is at most 250? Less than 250? More than 300? (Round your answers to five decimal places.)
at most 250 less than 250more than 300
(b)
What is the probability that a specimen's lifetime is between 100 and 250? (Round your answer to four decimal places.)
(c)
What value (in hr) is such that exactly 50% of all specimens have lifetimes exceeding that value? (Round your answer to three decimal places.)
hr
What are the pros and cons of a tubular heat exchanger?
An electric current of 32.30 A flows for 528. milliseconds. Calculate the amount of electric charge transported.
Be sure your answer has the correct unit symbol and the correct number of significant digits.
Chapter 3 Solutions
Principles of Instrumental Analysis
Ch. 3 - Prob. 3.1QAPCh. 3 - Prob. 3.2QAPCh. 3 - Prob. 3.3QAPCh. 3 - Prob. 3.4QAPCh. 3 - Prob. 3.5QAPCh. 3 - Prob. 3.6QAPCh. 3 - Prob. 3.7QAPCh. 3 - Prob. 3.8QAPCh. 3 - Prob. 3.9QAPCh. 3 - Prob. 3.10QAP
Ch. 3 - Prob. 3.11QAPCh. 3 - Prob. 3.12QAPCh. 3 - Prob. 3.13QAPCh. 3 - Prob. 3.14QAPCh. 3 - Prob. 3.15QAPCh. 3 - Prob. 3.16QAPCh. 3 - Prob. 3.17QAPCh. 3 - Prob. 3.18QAPCh. 3 - Prob. 3.19QAPCh. 3 - Prob. 3.20QAPCh. 3 - Prob. 3.21QAPCh. 3 - Prob. 3.22QAPCh. 3 - Prob. 3.23QAPCh. 3 - Prob. 3.24QAPCh. 3 - Prob. 3.25QAP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Mehuuul.ou9arrow_forwardWhich of the following relations describes the maximum energy that can be determined from the system at work at coinstant temperature? A dWmax = dA B) dWmax = dU - TdS C) dWmax = dQrev D dWmax = -SdT + VdP Increasing the supplied current in an electrolytic deposition process will A Allow liquid water to intercalate in between solid atoms B Dissolve the solid into the solution Decrease the amount of solid that will be deposited Decrease the time required to achieve desired thickness of the coatingarrow_forwardA2. Determine total differentials: dT = dT(s,v), dP = dP(s, v), dụ = dµ(s,v). %|arrow_forward
- Q4: The heater element of an electric kettle has a constant resistance of 120 Q and the applied voltage is 220 V. Calculate the time taken to raise the temperature of one liter of water from 15°C to 90°C assuming that 78% of the power input to the kettle is usefully employed. If the water equivalent of the kettle is 100 g, find how long will it take to raise a second liter of water through the same temperature range immediately after the first.arrow_forwardA 1.00 mol sample of water vapour is enclosed in a rigid container equipped with a 9.97 Ω electric resistance heater. When a current of 0.200 A is passed through the resistor for 100.0 s, the temperature of the apparatus was observed to increase by 0.465 K. Separate measurements on the evacuated container show that the heat capacity of the container and resistance heater is 60.50 J. K-1. Calculate the value of Cv for the gaseous water (water vapour)arrow_forward(2) If equal weights of polymer A and polymer B are mixed, Calculate M, and Mw of the m ixture Polymer A: M, = 35,000, Mw = 90,000 Polymer B: M, = 150,000, Mw = 300,000 72arrow_forward
- 2. Given three capacitors 4F, 6F, and 8F and a total electric potential of 150V, find all the following values if the capacitors is connected in a parallel. CT= C1- C2= C3= Q3= Qr= VT= Q1= Q2= V1= V2- V3=arrow_forward4.Eicosane is used as the internal standard. What would be a better internal standard and why?arrow_forwardCalculate the energy cost of an electroplating process running at 0.65 V and 48.9 A for 3149 s considering a cost of PhP52.96 per kWh? Round your answer to 2 decimal places.arrow_forward
- Solve it asap...arrow_forwardA current of 1.55 A from a 110 V source was passed through a heater for 8.5 min. The heater was immersed in a water bath. What quantity of energy was transferred to the water as heat?arrow_forwardPts) |Xtag), X2+ (aq) |/ y3+ *(aq) | Y(s) Given the line notation: where: x2+ + e x+ E° = 0.48 V y3+ + 3 e → Y E° = -2.35 V Pt4+ + 4 e - Pt E° = 1.20 V Calculate the EMF. Input values only with 2 decimal places. Do not include the unit. If positive value: DO NOT include the positive (+) sign If negative value: Please include the negative (-) signarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning