
Concept explainers
(a)
Interpretation:
Lewis structure of
Concept Introduction:
Lewis structure represents covalent bonds and describes valence electrons configuration of atoms. The covalent bonds are depicted by lines and unshared electron pairs by pairs of dots. The sequence to write Lewis structure of some molecule is given as follows:
- The central atom is identified and various other atoms are arranged around it. This central atom so chosen is often the least electronegative.
- Total valence electrons are estimated for each atom.
- single bond is first placed between each atom pair.
- The electrons left can be allocated as unshared electron pairs or as multiple bonds around
symbol of element to satisfy the octet (or duplet) for each atom. - Add charge on overall structure in case of polyatomic cation or anion.
Hybridization is calculated by the hybrid orbitals and to calculate hybrid orbitals we need to know the steric number that is given by,
The relation of steric number with geometry or shape is depicted in the table below.
(a)
Explanation of Solution
The molecule
Electronic configuration of
Electronic configuration of
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
Hence, 20 electrons are allocated as 3 lone pairs on each iodine atom and 1 lone pair on nitrogen. The Lewis structure of
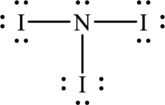
As from the structure above, there are 3 bond pairs and 1 lone pair on central atom.
Substitute 3 for number of atoms bonded to central atom and 1 for number of lone pairs on central atom in equation (1) to determine steric number.
The shape of the molecule is defined by the lone pairs present on the central atom. Molecule
(b)
Interpretation:
Lewis structure of
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
The molecule
Electronic configuration of
Electronic configuration of
Thus total valence electrons is sum of the valence electrons for each atom in
The skeleton structure
Hence, 18 electrons are allocated as 3 lone pairs on each iodine atom. The Lewis structure of
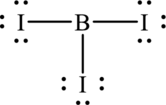
As from the structure above, there are 3 bond pairs and 0 lone pairs on central atom.
Substitute 3 for number of atoms bonded to central atom and 0 for number of lone pairs on central atom in equation (1) to determine steric number.
The shape of the molecule is defined by the lone pairs present on the central atom. Molecule
Want to see more full solutions like this?
Chapter 3 Solutions
SAPLINGPLUS ACCESS CHEMICAL PRINCIPLES
- Silane SiH4, phosphine (PH3), and hydrogen sulfide (H2S) melt at 185 C, 133 C, and 85 C, respectively. What does this suggest about the polar character and intermolecular attractions of the three compounds?arrow_forwardFind the planer density (Po) in (atom/mm) of (100) in a face centered cubic unit cell with lattice constant of 3.5 A, and determined the atomic radiusarrow_forwardExplain the concepts of viscosity, surface tension, and capillary action?arrow_forward
- What type(s) of intermolecular forces are expected between GeH4 molecules?arrow_forwardAre hydrogen bonds modelled in molecular mechanics as weak covalent bonds, as strong van der Waals or dispersion forces, or as electrostatic attractions?arrow_forwardCalculate the radius of a vanadium (V) atom, given that its density is 6.11 g/cm3 and that it crystallizes in a body-centred cubic unit cell.arrow_forward
- (a) What atoms must a molecule contain to participatein hydrogen bonding with other molecules of the samekind? (b) Which of the following molecules can formhydrogen bonds with other molecules of the same kind:CH3F, CH3NH2, CH3OH, CH3Br?arrow_forwardWhich has the lowest viscosity? C6 H14, C12H26, or C18H28arrow_forwardDefine the term Ion–Dipole Interactions?arrow_forward
- what is meant by the term polarizability?arrow_forwardWhat are the root mean square distances traveled (in one dimension) by a glycine molecule in water and by a sucrose molecule in water at 25°C in 1.0 s? (a) glycine molecule in water (Take D = 1.055 ✕ 10−9 m2·s−1.) (b) sucrose molecule in water (Take D = 5.216 ✕ 10−10 m2·s−1.)arrow_forwardliquid nitrogen has a density if 875.4 kg/m^3 at its normal boiling point. what volume does a ballon occupy at 1 atm and 298K of 3.1x 10^-3 L of liquid N3 is injected into it?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





