
ORGANIC CHEMISTRY TEXT PACKAGED - 2 YE
10th Edition
ISBN: 9781260024241
Author: Carey
Publisher: MCG/CREATE
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 32P
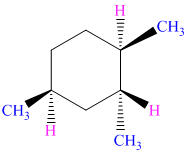
Write a structural formula for the most stable chair conformation of each of the following
compounds:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write structural formulas for the most stable conformation of each of the following compounds
cis-1-tert-Butyl-4-methylcyclohexane
Write the two chair conformations of each of the following and in each part designate which conformation would be more stable. Why?a) trans-1-tert-butyl-3-methylcyclohexaneb) trans-1-tert-butyl-4-methylcyclohexane
Provide the least stable chair conformation of cis-1-hydroxy-4-isopropylcyclohexane
Chapter 3 Solutions
ORGANIC CHEMISTRY TEXT PACKAGED - 2 YE
Ch. 3.1 - Identify the alkanes corresponding to each of the...Ch. 3.1 - Find the conformations in Figure 3.4 in which the...Ch. 3.2 - Sketch a potential energy diagram for rotation...Ch. 3.2 - Acetylcholine is a neurotransmitter in the central...Ch. 3.2 - Prob. 5PCh. 3.5 - The heats of combustion of ethylcyclopropane and...Ch. 3.8 - Prob. 7PCh. 3.10 - The following questions relate to a cyclohexane...Ch. 3.10 - Draw the most stable conformation of...Ch. 3.11 - Prob. 10P
Ch. 3.11 - Prob. 11PCh. 3.12 - Based on what you know about disubstituted...Ch. 3.12 - Write structural formulas for the most stable...Ch. 3.14 - Cubane (C4H8) is the common name of the polycyclic...Ch. 3.14 - Prob. 15PCh. 3.14 - Prob. 16PCh. 3.14 - Prob. 17PCh. 3.14 - Prob. 18PCh. 3.15 - Prob. 19PCh. 3 - Give the IUPAC names of each of the following: (a)...Ch. 3 - Draw Newman projections for the gauche and...Ch. 3 - Identify all atoms that are (a) anti and (b)...Ch. 3 - Prob. 23PCh. 3 - Prob. 24PCh. 3 - Prob. 25PCh. 3 - Prob. 26PCh. 3 - Prob. 27PCh. 3 - Prob. 28PCh. 3 - Oxidation of 4-tert-butylthiane proceeds according...Ch. 3 - The following are representations of two forms of...Ch. 3 - Draw (a) a Newman projection of the most stable...Ch. 3 - Write a structural formula for the most stable...Ch. 3 - Sight down the C-2-C-3 bond, and draw Newman...Ch. 3 - Prob. 34PCh. 3 - Sketch an approximate potential energy diagram for...Ch. 3 - Prob. 36PCh. 3 - Even though the methyl group occupies an...Ch. 3 - Which do you expect to be the more stable...Ch. 3 - Arrange the trimethylcyclohexane isomers shown in...Ch. 3 - Identify the more stable stereoisomer in each of...Ch. 3 - One stereoisomer of 1,1,3,5-tetramethylcyclohexane...Ch. 3 - One of the following two stereoisomers is...Ch. 3 - In each of the following groups of compounds,...Ch. 3 - The heats of combustion of the more and less...Ch. 3 - The measured dipole moment of ClCH2CH2Cl is 1.12D....Ch. 3 - Prob. 46PCh. 3 - Prob. 47PCh. 3 - Prob. 48DSPCh. 3 - Prob. 49DSPCh. 3 - Prob. 50DSPCh. 3 - Prob. 51DSPCh. 3 - Prob. 52DSPCh. 3 - Prob. 53DSP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Draw a Lewis structure for each of the following species: a. H2CO3 b. CO32 c. CH2O d. CO2
Essential Organic Chemistry (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following is a planar hexagon representation for one isomer of 1,2,4-trimethylcyclohexane. Draw the alternative chair conformations of this compound and state which of the two is more stable.arrow_forwardFollowing are structural formulas for 1,4-dioxane and piperidine. 1,4-Dioxane is a widely used solvent for organic compounds. Piperidine is found in small amounts in black pepper (Piper nigrum). (a) Complete the Lewis structure of each compound by showing all unshared electron pairs. (b) Predict bond angles about each carbon, oxygen, and nitrogen atom. (c) Describe the most stable conformation of each ring and compare these conformations with the chair conformation of cyclohexane.arrow_forwardConsider 1-bromo-2-methylpropane and draw the following. (a) The staggered conformation(s) of lowest energy (b) The staggered conformation(s) of highest energyarrow_forward
- Gibbs free energy differences between axial-substituted and equatorial-substituted chair conformations of cyclohexane were given in Table 2.4. (a) Calculate the ratio of equatorial to axial tert-butylcyclohexane at 25C. (b) Explain why the conformational equilibria for methyl, ethyl, and isopropyl substituents are comparable but the conformational equilibrium for tert-butylcyclohexane lies considerably farther toward the equatorial conformation.arrow_forwardmyo-Inositol, one of the isomers of 1,2,3,4,5,6-hexahydroxycyclohexane, acts as a growth factor in both animals and microorganisms. Draw the most stable chair conformation of myo-inositol.arrow_forwardThe diaxial conformation of cis-1, 3-dimethylcyclohexane is approximately 23 kJ/mol (5.4 kcal/mol) less stable than the diequatorial conformation. Draw the two possible chair conformations, and suggest a reason for the large energy difference.arrow_forward
- Given the structure of the following 1,2,3,5-tetrachlorocyclohexane: Draw the most stable chair conformation for the above 1,2,3,5-tetrachlorocyclohexane. Draw the least stable chair conformation for the above 1,2,3,5-tetrachlorocyclohexanearrow_forwardDraw the alternative chair conformations for the cis and trans isomers of 1,2-dimethylcyclohexane, 1,3-dimethylcyclohexane, and 1,4-dimethylcyclohexane. Q.) For which isomer(s) is one chair conformation more stable than the other?arrow_forwardProvide the least stable (highest energy) chair conformation of cis-1,3-dimethylcyclohexane.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License