
ORG CHEM LL W/ LL SG&CONPLUS PKG>IC<
5th Edition
ISBN: 9781260069228
Author: SMITH
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 3.3P
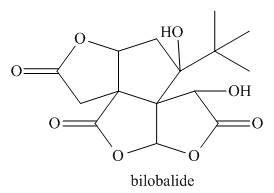
Classify a carbon atom by the number of carbons to which it is bonded can also be
done in more complex molecules that contain heteroatoms. Classify each
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the number of sp3-hybridized carbon atoms in each of these isomers.
1. (CH3)2 CO
2. CH3CH2CHO
Describe the structure of the SP2 hybridized carbon atom. With the participation of which orbital can it form a π-bond with another sp2-hybridized carbon atom?
Carbenes (CR2) are neutral compounds with divalent carbon atoms. Propose two different ways that carbenes could exist. Provide the hybridization of each carbene carbon and predict the bond angles based on what you know about hybridization
Chapter 3 Solutions
ORG CHEM LL W/ LL SG&CONPLUS PKG>IC<
Ch. 3 - Prob. 3.1PCh. 3 - (a) Classify the carbon atoms in each compound as...Ch. 3 - Problem 3.3 Classify a carbon atom by the number...Ch. 3 - Classify each alkyl halide and alcohol as , or...Ch. 3 - Prob. 3.5PCh. 3 - Prob. 3.6PCh. 3 - Draw the structure of a compound of molecular...Ch. 3 - Prob. 3.8PCh. 3 - Prob. 3.9PCh. 3 - Draw the structure of a compound fitting each...
Ch. 3 - Draw structures that fit each description and name...Ch. 3 - What types of intermolecular forces are present in...Ch. 3 - Which compound in each pair has the higher boiling...Ch. 3 - Explain why the boiling point of propanamide, is...Ch. 3 - Predict which compound in each pair has the higher...Ch. 3 - Prob. 3.16PCh. 3 - Which compounds are water soluble? a. b. c.Ch. 3 - a Label the hydrophobic and hydrophilic portions...Ch. 3 - Prob. 3.19PCh. 3 - Prob. 3.20PCh. 3 - Prob. 3.21PCh. 3 - Prob. 3.22PCh. 3 - Problem 3.23 (a) What types of intermolecular...Ch. 3 - Prob. 3.24PCh. 3 - Prob. 3.25PCh. 3 - Problem 3.26 Label the electrophilic and...Ch. 3 - Problem 3.27 Considering only electron density,...Ch. 3 - Prob. 3.28PCh. 3 - 3.29
Identify the functional groups in the...Ch. 3 - Prob. 3.30PCh. 3 - 3.31 For each alkane: (a) classify each carbon...Ch. 3 - 3.32 Identify the functional groups in each...Ch. 3 - 3.33 Identify each functional group located in the...Ch. 3 - 3.34 (a)Identify the functional groups in...Ch. 3 - Draw seven constitutional isomers with molecular...Ch. 3 - Prob. 3.36PCh. 3 - Prob. 3.37PCh. 3 - Prob. 3.38PCh. 3 - Intramolecular force of attraction are often...Ch. 3 - 3.40 (a) Draw four compounds with molecular...Ch. 3 - 3.41 Rank the compounds in each group in order of...Ch. 3 - Explain why CH3CH2NHCH3 has higher boiling point...Ch. 3 - Prob. 3.43PCh. 3 - 3.44 Rank the following compounds in order of...Ch. 3 - Prob. 3.45PCh. 3 - 3.46 Rank the following compounds in order of...Ch. 3 - 3.47 Which of the following molecules can hydrogen...Ch. 3 - 3.48 Explain why diethylether and have similar...Ch. 3 - Prob. 3.49PCh. 3 - 3.50 Predict the solubility of each of the...Ch. 3 - Prob. 3.51PCh. 3 - Prob. 3.52PCh. 3 - 3.53 THC is the active component in marijuana, and...Ch. 3 - Prob. 3.54PCh. 3 - Prob. 3.55PCh. 3 - 3.56 Label the electrophilic and nucleophilic...Ch. 3 - 3.57 By using only electron density arguments,...Ch. 3 - 3.58 The composition of a cell membrane is not...Ch. 3 - Prob. 3.59PCh. 3 - 3.60 Quinapril (trade name Accupril) is a drug...Ch. 3 - 3.61 Answer each question about oxycodone, a...Ch. 3 - Prob. 3.62PCh. 3 - Prob. 3.63PCh. 3 - 3.64 Explain why A is less water soluble than B,...Ch. 3 - 3.65 Recall from section 1.10B that there is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Classifying a carbon atom by the number of carbons to which it is bonded can also be done in more complex molecules that contain heteroatoms. Classify each sp3 hybridized carbon atom in bilobalide, a compound isolated from Ginkgo biloba extracts, as 1°, 2°, 3°, or 4°.arrow_forwardRedraw compound 2.1 and label each carbon atom and the oxygen atom with its hybridisationarrow_forwardhow many sp2 hybridized carbon?arrow_forward
- Pyrethrins, such as jasmolin II(below), are a group of naturalcompounds synthesized by flowers of the genus Chrysanthemum(known as pyrethrum flowers) to act as insecticides.(a) Circle and name the functional groups in jasmolin II.(b) What is the hybridization of the numbered carbons?(c) Which, if any, of the numbered carbons are chiral centers?arrow_forwardWhat kind of hybridization do you expect for each carbon atom in the molecule? a.1-Buten-3-ynearrow_forwardDraw structural formulas for all linear (not ring) constitutional isomers that have the molecular formula C4H4 and indicate which of the following they possess: 1. sp hybridized carbon atoms 2. sp2 hybridized carbon atoms 3. sp3 hybridized carbon atoms 4. sigma bonds, pi bondsarrow_forward
- Draw electron configuration diagrams for carbon in an unhybridized, sp3 -hybridized state, sp2 -hybridized state, and sp-hybridized state.arrow_forwardProstaglandin F2α, a hormone that causes uterine contraction during childbirth, has the following structure. Are the two hydroxyl groups (-OH) on the cyclopentane ring cis or trans to each other? What about the two carbon chains attached to the ring?arrow_forwardZingerone gives ginger its pungent taste. How many sp2 hybridized carbons are present?arrow_forward
- Classifying a carbon atom by the number of carbons to which it is bonded can also be done in more-complex molecules that contain heteroatoms. Classify each sp3 hybridized carbon atom in bilobalide, a compound isolated from Ginkgo biloba extracts, as 1°, 2°, 3°, or 4°.arrow_forwardWhich compound contains an sp2-hybridized carbon atom?arrow_forwardVancomycin is a useful antibiotic for treating infections in cancer patients on chemotherapy and renal patients on dialysis. How many amide functional groups are present in vancomycin? Which OH groups are bonded to sp3 hybridized carbon atoms and which are bonded to sp2 hybridized carbons?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
ENVIRONMENTAL POLLUTION; Author: 7activestudio;https://www.youtube.com/watch?v=oxtMFmDTv3Q;License: Standard YouTube License, CC-BY