
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
9th Edition
ISBN: 9780137249442
Author: Wade
Publisher: INTER PEAR
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 3.52SP
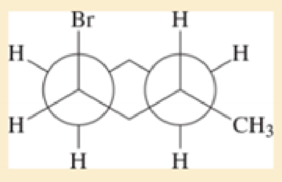
This is a Newman projection of a substituted cyclohexane.
- a. Draw the equivalent chair form.
- b. Draw the equivalent structure using wedge and dash notation on a cyclohexane hexagon.
- c. Give the IUPAC name.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the structure corresponding to each IUPAC name.
a. 2,3,4,5−tetramethyldecane
draw structure ...
b. cyclononane
draw structure ...
B. For each write the correct IUPAC name
The following Newman projection corresponds to what IUPAC name?
H
H.
.CH2CH3
CH3CH2
H.
H
Chapter 3 Solutions
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
Ch. 3.2 - Using the general molecular formula for alkanes:...Ch. 3.3B - Name the following alkanes and haloalkanes. When...Ch. 3.3B - Write structures for the following compounds. a....Ch. 3.3B - Provide IUPAC names for the following compounds....Ch. 3.3B - Prob. 3.5PCh. 3.3B - Prob. 3.6PCh. 3.3B - Prob. 3.7PCh. 3.3B - Draw the structures of the following compounds. a....Ch. 3.3B - Without looking at the structures, give molecular...Ch. 3.4C - Prob. 3.10P
Ch. 3.7B - Prob. 3.11PCh. 3.7C - Draw a graph similar to Figure 3-9, of the...Ch. 3.8B - Draw a graph similar to Figure 3-11, of the...Ch. 3.9 - Draw a perspective representation of the most...Ch. 3.10C - Give IUPAC names for the following compounds.Ch. 3.10C - Draw the structure and give the molecular formula...Ch. 3.11 - Which of the following cycloalkanes are capable of...Ch. 3.11 - Give IUPAC names for the following cycloalkanes.Ch. 3.12B - The heat of combustion of...Ch. 3.12C - Prob. 3.20PCh. 3.13B - The cyclohexane chair shown in Figure 3-22 has the...Ch. 3.13B - Draw 1,2,3,4,5,6-hexamethylcyclohexane with all...Ch. 3.14 - Prob. 3.23PCh. 3.14 - Prob. 3.24PCh. 3.14 - Draw the most stable conformation of a....Ch. 3.15 - Prob. 3.26PCh. 3.15 - a. Draw both chair conformations of cis-1...Ch. 3.15 - Prob. 3.28PCh. 3.15A - Draw the two chair conformations of each of the...Ch. 3.15B - Draw the most stable conformation of a....Ch. 3.16A - Name the following compounds.Ch. 3.16B - Prob. 3.32PCh. 3 - Prob. 3.33SPCh. 3 - Prob. 3.34SPCh. 3 - 3-35 a. Draw and name the five cycloalkane...Ch. 3 - Draw the structure that corresponds with each...Ch. 3 - Each of the following descriptions applies to more...Ch. 3 - Write structures for a homologous series of...Ch. 3 - Prob. 3.39SPCh. 3 - Construct a graph, similar to Figure 3-11, of the...Ch. 3 - The following names are all incorrect or...Ch. 3 - In each pair of compounds, which compound has the...Ch. 3 - There are eight different five-carbon alkyl...Ch. 3 - Use a Newman projection about the indicated bond...Ch. 3 - a. Draw the two chair conformations of...Ch. 3 - Draw the two chair conformations of each compound,...Ch. 3 - Using what you know about the conformational...Ch. 3 - Prob. 3.48SPCh. 3 - Draw Newman projections along the C3C4 bond to...Ch. 3 - Prob. 3.50SPCh. 3 - The most stable form of the common sugar glucose...Ch. 3 - This is a Newman projection of a substituted...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Name using IUPAC substitutive nomenclature nules A. В. 2. Circle the molecules that are the meso-fonn trans-1.2-dimethykyclohexane cis-1.3-dimethylcyclonexane trans-1,4-dimethyleyclohexanearrow_forwardBe sure to answer all parts. Draw the structure corresponding to each IUPAC name. a. 6-butyl-3-methyldecane CH₂ edit structure ... b. 4,4,5,5-tetramethylnonane X H₂C₂ CH₂ edit structure ...arrow_forwardWhat is the IUPAC name of the following compound? a. trans-1-isopropyl-4-methylcyclopentane b. cis-1-tert-butyl-2-methylcyclopentane c. trans-1-tert-butyl-2-methylcyclopentane d. cis-1-isopropyl-2-methylcyclopentane a O barrow_forward
- Be sure to answer all parts. Draw the structure corresponding to each IUPAC name. a. 3,3-dimethylpentane CH₂ CH₂ edit structure... b. 3-ethyl-4-methylhexane -CH₂ CH₂ CH3 CH₂ edit structure ... CH₂ Xarrow_forwardConsider the synthesis scheme in Figure 12. Give the preferred IUPAC name of C? [Use lowercase letters. Do not use spaces if it is not required in the name.] *arrow_forwarda.) Draw B as a hexagon with wedges and dashed wedges to show the stereochemistry of substituents.b.) Draw a stereoisomer of A as a hexagon using wedges and dashed wedges to show the orientation of substituents.arrow_forward
- 2. Draw structures corresponding to the each name. a. 5,6-dimethylhept-2-yne b. cis-1-ethynyl-2-methylcyclopentane 5-tert-butyl-6,6-dimethylnon-3-yne C.arrow_forwardGlucose is a simple sugar with five substituents bonded to a sixmembered ring. a.) Using a chair representation, draw the most stable arrangement of these substituents on the six-membered ring.b.) Convert this representation to one that uses a hexagon with wedges and dashed wedges.c.) Draw a constitutional isomer of glucose.d.) Draw a stereoisomer that has an axial OH group on one carbonarrow_forward1. Draw the structure for each compound. a.(3R)-3-methylhexane b. (3R,5S,6R)-5-ethyl-3,6-dimethylnonanearrow_forward
- Consider the synthesis scheme in Figure 12. Give the preferred IUPAC name of C? [Use lowercase letters. Do not use spaces if it is not required in the name.] * Your answerarrow_forward• Question 6: Which alkene in each pair is more stable? a. b. CH3CH₂ H C=C or CH₂CH3 H or CH3CH₂ H H CH₂CH3 2 C. CH3 or CH3arrow_forwardWhat is the IUPAC name? CH, a. (S)-4-methylpent-1-ynal b. (R)-2-methylpent-4-yank c. (S)-2-methylpent-4-yank d. (R)-4-methylpent-1-ynalarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License