
Concept explainers
PRACTICE PROBLEM 3.3
Which of the following are potential Lewis acids and which are potential Lewis bases?
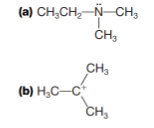
(c)
(d)
(e)
(f)
Trending nowThis is a popular solution!

Chapter 3 Solutions
EBK ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Chemistry: The Molecular Nature of Matter
Chemistry: A Molecular Approach (4th Edition)
Chemistry (7th Edition)
Inorganic Chemistry
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
- Consider the following bases: A Which of the following statements is true? (i) The ions increase in basicity as follows: A < B < C, with C being the strongest base. (ii) The conjugate acid of C would have the highest pKa of the conjugate acids of the three bases. (iii) A is the best electron donor. O ii only O i only O two of i, ii and i O i only O None of i, ii and iiiarrow_forwardSolve the following Given that pH = 13, 7.5 , 7.4 find: (a) pOH (b) H3O+ (c) OH-arrow_forwardIdentify the Lewis acid and Lewis base in each equation:arrow_forward
- For the following five reactions: (i) Cl− + AlCl3 → AlCl4−(ii) (CH3)3N + HCl → (CH3)3NHCl(iii) OH− + H3O+ → 2H2O(iv) Co3+ + 6NH3 → Co(NH3)6(v) F− + SiF4 → SiF5− Identify the Lewis acids. Choose from the list below and enter the five letters in order. (e.g. ACEGI, BDFHJ, etc.) A) Cl− B) AlCl3 C) (CH3)3N D) HCl E) OH− F) H3O+ G) Co3+ H) NH3 I) F− J) SiF4arrow_forward(4) Which acid below would have a stronger conjugate base? Give the reason (a) HCN, Ka 6.2 x 10-10 Ka=2.3 x 10-⁹ (b) HBrO,arrow_forwardFor each conjugate acid-base pair, identify the first species as an acid or a base and the second species as its conjugate acid or base. In addition, draw Lewis structures for each species, showing all valence electrons and any formal charge. (a) HCOOH HCOO-arrow_forward
- Which is the stronger base, (CH3)3N or H2BO3− and why?arrow_forwardBenzoic acid (C6H5COOH) and aniline (C6H5NH2) areboth derivatives of benzene. Benzoic acid is an acid withKa = 6.3 x10-5 and aniline is a base with Ka = 4.3 x10-10. (a) What are the conjugate base of benzoic acid andthe conjugate acid of aniline? (b) Anilinium chloride(C6H5NH3Cl) is a strong electrolyte that dissociates intoanilinium ions (C6H5NH3+) and chloride ions. Which willbe more acidic, a 0.10 M solution of benzoic acid or a 0.10M solution of anilinium chloride? (c) What is the value ofthe equilibrium constant for the following equilibrium?C6H5COOH(aq) + C6H5NH2(aq) ⇌ C6H5COO-(aq) + C6H5NH3+(aq)arrow_forwardConsider two acids: HCO2H (formic acid, pKa = 3.8) and pivalic acid [(CH3)3CCO,H, pK = 5.0]. (a) Which acid has the larger K? (b) Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base? (d) When each acid is dissolved in water, for which acid does the equilibrium lie further to the right? %3D The pKa values in Table 2.1 span a large range (-7 to 50). The pK, scale is logarithmic. small difference in pK, translates into a large numerical difference, For example, the diffe between the pK, of NH3 (38) and CH2=CH, (44) is six pKa units. This means that NH, is one million times more acidic than CH,=CH,.arrow_forward
- For the following five reactions: (i) Cl− + AlCl3 → AlCl4−(ii) (CH3)3N + HCl → (CH3)3NHCl(iii) OH− + H3O+ → 2H2O(iv) Co3+ + 6NH3 → Co(NH3)6(v) F− + SiF4 → SiF5− Identify the Lewis bases. Choose from the list below and enter the five letters in order. (e.g. ACEGI, BDFHJ, etc.) A) Cl− B) AlCl3 C) (CH3)3N D) HCl E) OH− F) H3O+ G) Co3+ H) NH3 I) F− J) SiF4arrow_forward(a) Calculate [OH-] of a 4.40×10-1 M aqueous solution of triethylamine ((C2H5)3N, Kb = 4.0×10-4). (b)Calculate the pH of the above solution.arrow_forward3. For each of the following pairs of ions, identify the stronger base: (a) OH or NHz (b) F or Ch (c) CHSCH2O or CHSCOO Answer:arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
