
Interpretation:
The meaning of electroosmotic flow and the reason for itsoccurrence needs to be explained.
Concept introduction:
When electric field is applied in a liquid, the ions present in the liquid moves under the influence of potential gradient. This flow of liquid is termed as electroosmotic flow. It is most obvious is those having small channels. It occurs in buffered solutions and natural water.
Answer to Problem 30.1QAP
Electroosmotic flow of liquid is caused by gradient in potential across the capillary tube on the flowing liquid that has dissolved ions. This is caused when the capillary walls get electrically charged.
Explanation of Solution
When a capillary tube is filled with fluid having dissolved ions, and electrical field is applied, the buffer ions move to respond to the electrical charges. As the solvent which is water is neutral, it remains stationary and the fluid ions move towards its electrodes with respect to the charges applied. Below is depiction of movement of electrons:
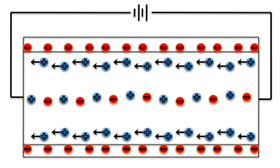
There is coulombic force induced by charges of ions in the solution wherein its net mobile charge causes electroosmotic flow. During this process of maintaining
Electroosmotic flow of liquid is caused due to a potential gradient across the capillary tube on the flowing liquid containing dissolved ions. This is caused when the capillary walls get electrically charged due to the application of electric field from outside.
Want to see more full solutions like this?
Chapter 30 Solutions
Principles of Instrumental Analysis, 6th Edition
- For weak electrolytes, such as acetic acid, at very low concentrations the molar conductivity drops rapidly as the concentration increases. However, at higher concentration, there’s only a slight decrease in the molar conductivity. True Falsearrow_forward4. In the water and glycerol mixture, the relationship between molar volume and mol fraction of glycerol is given in the following table at 20°C: X₂ (mol fraction of glycerol) 0 Molar volume (cm³ mol-¹) 18.05 19.18 20.53 24.18 0.0212 0.0466 0.1153 0.2269 0.4390 55.87 0.6923 1 73.02 (a) Calculate the partial molar volume of water and glycerol as a function of X2. (b) Show that the molar volume in the table can be calculated using the partial molar volume. 30.21 41.82arrow_forwardWhich of the following statement/s is/are TRUE? The Arrhenius Theory cannot be applied to account for the behavior of the molar conductivity of strong electrolytes, since these solutes are completely dissociated, at least in dilute solutions. Debye=Huckel Theory of interionic attraction satisfactorily explains the variation of the molecular conductivity of strong electrolytes with concentration. The ions interact with the solvent molecules through dipole-dipole interaction. When an electric potential is applied across the solution, the ions migrate and the ionic atmosphere around these ions is modified.arrow_forward
- Which of the following statement/s is/are TRUE? The Arrhenius Theory cannot be applied to account for the behavior of the molar conductivity of strong electrolytes, since these solutes are completely dissociated, at least in dilute solutions. Debye=Huckel Theory of interionic attraction satisfactorily explains the variation of the molecular conductivity of strong electrolytes with concentration. The ions interact with the solvent molecules through dipole-dipole interaction. When an electric potential is applied across the solution, the ions migrate and the ionic atmosphere around these ions is modified. I, II, IV I, III II, III, IV I, II I, II, IIIarrow_forwardWhen pure furfural (solvent) is added to a mixture containing 0.2 mass fraction diphenylhexane and 0.8 mass fraction docosane, two separate layers can be obtained. If the composition of the two layers are as shown in the table, how much solvent was added to 500 kg of solution of docosane and diphenylhexane?arrow_forward(c) Are the strengths of the interactions between the particles in the solute and between the particles in the solvent before the solute and solvent are combined greater than, less than, or equal to the strengths of the interactions between solute particles and solvent particles after dissolution? Explain.arrow_forward
- The phase diagrams for a pure solvent and the solvent in a solution are shown. Identify the normal freezing (fpsoly) and boiling (bpsolv) points for the pure solvent and the normal freezing (fpsoln) and boiling (bpgoln) points of the solution at 1 atm. Assume the solute is nonvolatile and that the solid that freezes from solution is pure solvent. 1 atm Liquid Solid Answer Bank fpsolv bpsolv fpsoln bpsoln Gas Temperature (°C) Pressure (atm)arrow_forwardAICI, ? reflux + O=arrow_forwardwhat are the diffrences between strong and weak electrolytes ?arrow_forward
- A standard solution is prepared by diluting the stock solution and used in chemical analysis for preparing the standard curve. Larutan piawai disediakan dengan melakukan pencairan larutan stok dan digunakan dalam analisis kimia untuk penyediaan lengkuk piawai. A) Illustrate how do you prepare a stock solution of 0.01 g of NaNO2 in one liter of distilled water in laboratory.arrow_forwardKMnO4 and Na2C2O4 solutions were used in the reactions that took place in a back titration to determine the amount of H2O2 in a sample. Calculate the concentration of H2O2 in the sample (w / v) as% by making appropriate assumptions for the volumes and normality of all these solutions.arrow_forwardAs solute is dissolved in a solvent, the vapor pressure of the solution changes according to Raoult's law Psoln Psolv X Xsolv where Psoln is the vapor pressure of the solution, Psolv is the vapor pressure of the pure solvent, and Xsolv is the mole fraction of the solvent. If the solute dissociates into ions, the term Xsolv must be modified to take into consideration the total number of moles of particles in the solution, both ions and molecules. When a solution contains two volatile components, A and B, the total pressure of the solution is equal to the sum of the individual vapor pressures according to Dalton's law as follows: Ptotal PAX XA +PB X XB Part A At 55.0 °C, what is the vapor pressure of a solution prepared by dissolving 75.8 g of LiF in 253 g of water? The vapor pressure of water at 55.0 °C is 118 mmHg. Assume complete dissociation of the solute. Express your answer to three significant figures and include the appropriate units. ► View Available Hint(s) μA Value Submit…arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,

