
Concept explainers
Draw the products formed when triacylglycerol A is treated with each reagent. Rank compounds A, B, and C in order of increasing melting point.
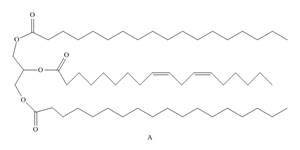 a.
a.
b.
c.
Want to see the full answer?
Check out a sample textbook solution
Chapter 30 Solutions
Organic Chemistry -Study Guide / Solution Manual (Custom)
- How are phospholipids similar in structure to a soap molecule? What structural difference accounts for the fact that phospholipids form lipid bilayers rather than the micelles that soaps form?arrow_forwardDraw the general block diagram for a triacylglycerol.arrow_forward21-100 What are the functions of a cell membrane? To what extent is a bilayer that consists entirely of lipids able to carry out these functions?arrow_forward
- Describe the structure of a micelle formed by the association of fatty acid molecules in water. What forces hold the micelle together?arrow_forward1. What is the difference between fats and oils? 2. Explain the difference between saturated and unsaturated triglycerides? 3. What is the difference between a monounsaturated and a polyunsaturated fat? 4. Which is more beneficial, saturated or unsaturated fatty acids? Why? 5. What are the functions of glycerides?arrow_forwardList the intermolecular attractions that occur betweena water molecule and a glycerol molecule.arrow_forward
- Here are several (hypothetical) values of the % saturated fatty acyl chains in samples of triglycerides. % saturated FA chains Sample W. 15 Sample X 30 Sample Y 45 Sample Z 60 Let's suppose the temperature in our cellar was 20 degrees C. Which of the samples would be liquid when stored in the cellar? Group of answer choices Sample W Sample X Sample Y Sample Zarrow_forwardHow do I saponify 3 mixed unsaturated triglyceride with 3NaOH?arrow_forwardYou are given 150 mL of an oil made up of a mixture of 3 fatty acids: palmitoleic acid (C 16:1 (n-7)) erucic acid (C 22:1 (n-9)) stearidonic acid (C 18:4 (n-3)) You know that the 150 mL sample of this oil was prepared by adding 60 mL of palmitoleic acid to 90 mL of a mixture of erucic acid and stearidonic acid. Using a hydrometer you carefully measure the density of the 150 mL sample of oil and your analysis indicates that it has a density of 0.900 g/cm3. The densities of the individual fatty acids are: palmitoleic acid = 0.894 g/cm3 erucic acid = 0.860 g/cm3 stearidonic acid = 0.9334 g/cm3 Can you determine the volumes (in mL) of erucic acid and stearidonic acid in the total volume (150 mL) of the oil mixture?arrow_forward
- 1.What are the products of the reaction between a fat and sodium hydroxide? 2.Explain how soap can remove non-polar substances from surfaces. 3.What is the reaction used to make soap?arrow_forwardA H-bond has a delta u of around 1 kcal/mol. Answer both part a and part b please. Part a) What fraction of the hydrogen bonds would be broken at 27 celsius (room temperature) and how many hydrogen bonds would be broken at 100 celsius? Part b) Using your answer from Part a, explain why proteins are stored on ice?arrow_forward4. Which of the following best describe the physical properties of fatty acids?a. All physical properties of fatty acids are influenced by the length and degreeof unsaturation of their carbon chain.b. Melting points of fatty acids are influenced by the carbon chain length.c. Melting points of fatty acids are influenced by their degree of unsaturation.d. Alltheabove.5. Triacylglycerols are ________.a. insolubleinwater.b. partially soluble in water.c. soluble in water at elevated temperatures.d. solubleinwater.6. The backbone of a triglyceride is __________.a. a three-carbon molecule with an aldehyde and two hydroxyl groups.b. a three-carbon molecule with three hydroxyl groups.c. esterified fatty acids fused with 1 to 3 carbons.d. threelong-chainfattyacids.7. Triacylglycerols are _____ in their ability to store energy when compared to glycogen, the energy storing form of glucose in the human body.a. equally efficientb. insignificantc. less efficientd. more efficient 8. The essential…arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





