
Interpretation: The shorthand structures of poly(vinyl chloride) and nylon
Concept introduction: The molecules which are obtained by the combination of similar units and possessing high molecular mass are known as
Answer to Problem 31.1P
The shorthand structures of poly(vinyl chloride) and nylon
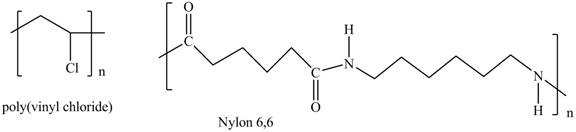
Explanation of Solution
The monomer required for the formation of poly(vinyl chloride) is vinyl chloride.
A shorthand structure of polymers represents the brackets that are placed around the repeating unit of the chain.
The shorthand structure of poly(vinyl chloride) and nylon
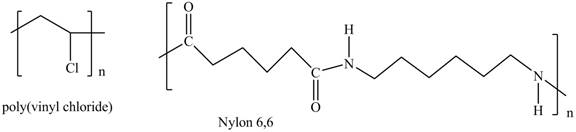
Figure 1
The shorthand structures of poly(vinyl chloride) and nylon
Want to see more full solutions like this?
Chapter 31 Solutions
KCTCS Organic Chemistry Value Edition (Looseleaf) - Text Only
- what is the polyESTER version of nylon-6,10, and what monomers make the polyester?arrow_forwardHydrogen bonding between polyamide chains plays animportant role in determining the properties of a nylonsuch as nylon 6,6 (Table 12.6). Draw the structuralformulas for two adjacent chains of nylon 6,6 and showwhere hydrogen-bonding interactions could occur betweenthem.arrow_forwardWhat do the two numbers correspond to in the name “Nylon-6,10?” Write the chemical structure of the repeat unit for “Nylon-4,6".arrow_forward
- 5. 1) Discuss how polymerization changes the properties of any ensemble of amino acids so that a protein polymer functions essentially differently than an aggregate of amino acids. 2) Why does a protein need to be a polymer in order to properly function??arrow_forwardWhich factor imparts crystalline nature to a polymer like nylon?arrow_forwardBriefly Differentiate between Tg and Tm polymers.arrow_forward
- Why is sulfur mustard comparatively less permeable to urethane polymer than polymers like polyethylene. Will a polyethylene gas mask be effective against sulfur mustard?arrow_forwardWhat is the structural difference between HDP and LDP? How does the structure account for different behaviour and nature, hence the use of a polymer?arrow_forwardPolystyrene synthetic polymer Monomer - styrene It is clear,hard,brittle and rigid Use- rigid trays , container, disposable eating utensils,foamed cups,plates and bowls. Briefly explain the point above about why Polystyrene is a synthetic polymer.arrow_forward
- Novolac is the linear polymer which on heating with formaldehyde forms cross-linked bakelite. Write the structures of monomers and the polymer novolac.arrow_forwardBriefly describe, using examples and sketches as appropriate, an experiment that illustrates how self-assembly can be used as a “one pot” synthesis of supramolecular polymer micellesarrow_forwardA natural polymer has been made called polylactic acid. What would be the name of the monomer?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning




