
One component of jojoba oil is a wax formed from eicosenoic acid
Interpretation: The structure of the wax formed from eicosenoic acid
Concept introduction: Waxes are lipids, which are hydrolysable. They contain ester
Answer to Problem 31.1P
The structure of the wax formed from eicosenoic acid
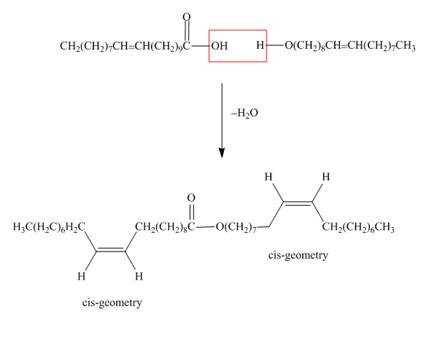
Figure 1
Explanation of Solution
Waxes are lipids, which are hydrolysable. They contain ester
The treatment of eicosenoic acid
The structure of the wax formed from eicosenoic acid
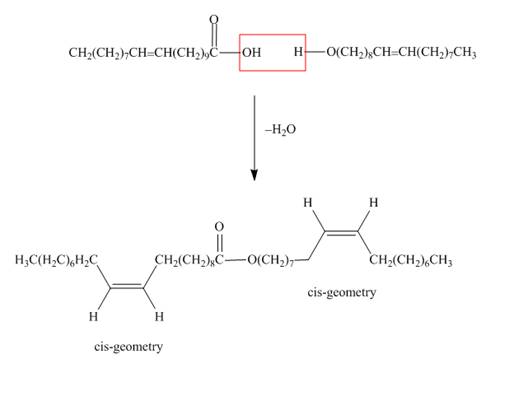
Figure 1
The structure of the wax formed from eicosenoic acid
Want to see more full solutions like this?
Chapter 31 Solutions
ORGANIC CHEMISTRY(LL)W/ACCESS>CUSTOM<
- Biological waxes have a head and two tails structure. Give the chemical identity of the head and of the two tails.arrow_forwardTrue/False - Waxes are different from fats and oils in that they are simple monoesters with long saturated carbon chains. A. True B. Falsearrow_forwardWaxes belong to which class of compounds? esters alcohols carboxylic acids ethersarrow_forward
- Draw the skeletal structures of the hydrolysis products carboxylic acid and alcohol formed when the wax is treated with aqueous sulfuric acid. CH3(CH2)16COO(CH2)17CH3arrow_forwardwhat functional group ch2ch3 is?arrow_forwardOf the alcohols with the molecular formula C4H9OH, which has the highest boiling point?arrow_forward
- Propene (CH 3CH = CH 2) can be formed by dehydrating two different alcohols. Draw the structures of both alcohols.arrow_forwardDraw the structure of a wax formed from stearic acid [CH3(CH2)16COOH] and the alcohol.arrow_forwardDraw the structure of the 1 isomer of C8H18 that contains 4 methyl branches on the main chain.arrow_forward
- 1a. . Isomers are responsible for the diversity of organic compounds. true or false? b. Positional isomers are molecules with different arrangements of the carbon skeleton but with the same chemical formula. true or false? c.)arrow_forwardDraw a structural formula of the product of oxidation of natural rubber by ozone followed by a workup in the presence of (CH3)2S. Name each functional group present in this product.arrow_forwardHow would these chemicals react with (CH2=CH)2CuLi?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





