
Concept explainers
Locate the isoprene units in each compound.
a. 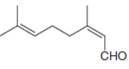 e.
e. 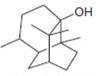 g.
g. 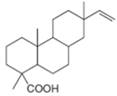
neral patchouli alcohol dextropimaric acid
b. 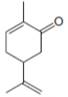 f.
f. 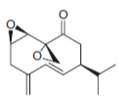 h.
h.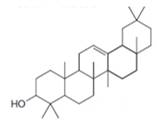
carvone periplanone B
c. 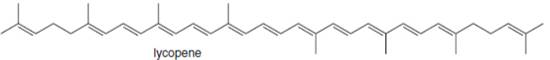
d. 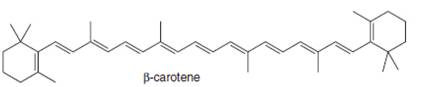
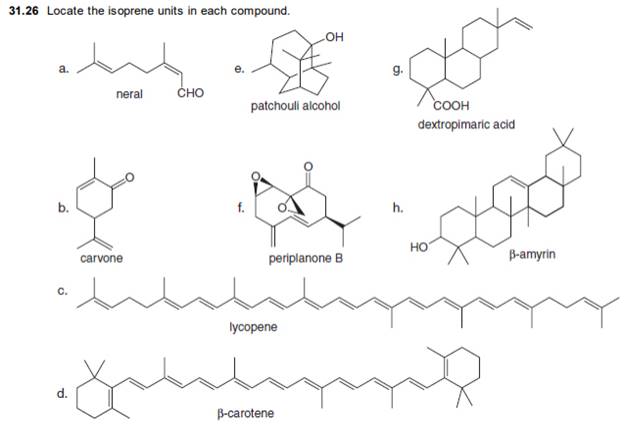
(a)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the neral are highlighted by blue color as shown below.
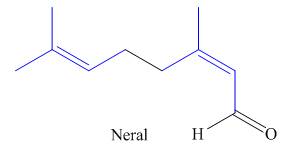
Figure 1
Explanation of Solution
The structure of isoprene is,
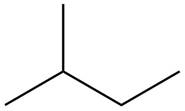
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 1
The highlighted bonds represent the isoprene unit. There are two isoprene units present in the given compound.
The isoprene units in the neral are shown in Figure 1.
(b)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the carvone are highlighted by blue color as shown below.
Figure 3
Explanation of Solution
The structure of isoprene is,
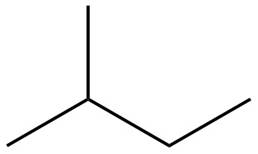
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 3
The highlighted bonds represent the isoprene unit. There are two isoprene units present in the given compound.
The isoprene units in the carvone are shown in Figure 3.
(c)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the lycopene are highlighted by blue color as shown below.
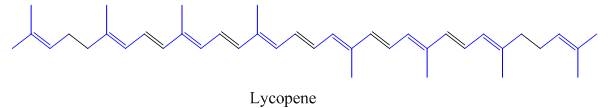
Figure 4
Explanation of Solution
The structure of isoprene is,
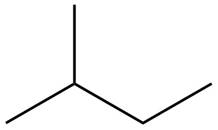
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are are highlighted by blue color as shown below.

Figure 4
The highlighted bonds represent the isoprene unit. There are eight isoprene units present in the given compound.
The isoprene units in the lycopene are shown in Figure 4.
(d)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the
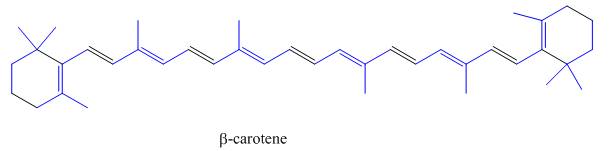
Figure 5
Explanation of Solution
The structure of isoprene is,
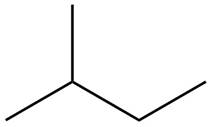
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.

Figure 5
The highlighted bonds represent the isoprene unit. There are eight isoprene units present in the given compound.
The isoprene units in the
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the patchouli alcohol are highlighted by blue color as shown below.
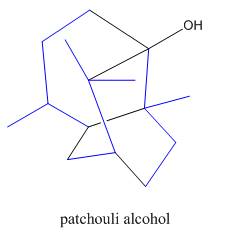
Figure 6
Explanation of Solution
The structure of isoprene is,
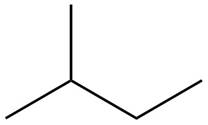
Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are are highlighted by blue color as shown below.

Figure 6
The highlighted bonds represent the isoprene unit. There are three isoprene units present in the given compound.
The isoprene units in the patchouli alcohol are shown in Figure 6.
(f)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the periplanone B are highlighted by blue color as shown below.
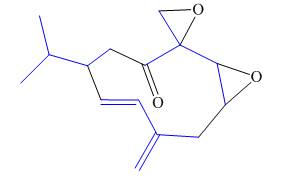
Figure 7
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are highlighted by blue color as shown below.
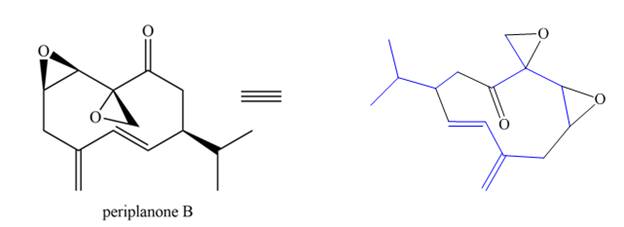
Figure 7
The highlighted bonds represent the isoprene unit. There are three isoprene units present in the given compound.
The isoprene units in the periplanone B are shown in Figure 7.
(g)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the dextropimaric acid are highlighted by blue color as shown below.
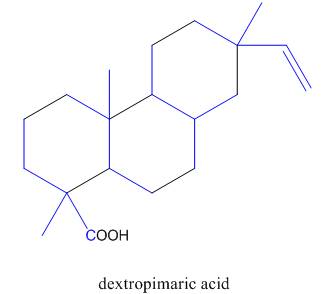
Figure 8
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are shown below.

Figure 8
The highlighted bonds represent the isoprene unit. There are four isoprene units present in the given compound.
The isoprene units in the dextropimaric acid are shown in Figure 8.
(h)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: Terpenes are naturally occurring compounds that are present in plants and animals. Terpenes contain one or more isoprene units. Terpenoids are derivatives of terpenes with oxygen containing functional group such as carbonyl groups.
Answer to Problem 31.26P
The isoprene units in the
Figure 9
Explanation of Solution
The structure of isoprene is,

Figure 2
The isoprene unit contains four carbon atoms in the long chain and one carbon atom in branch. Isoprene unit in a terpene consists of carbon-carbon sigma or pi bonds.
The isoprene units in the given compound are shown below.
Figure 9
The highlighted bonds represent the isoprene unit. There are four isoprene units present in the given compound.
The isoprene units in the
Want to see more full solutions like this?
Chapter 31 Solutions
ORGANIC CHEMISTRY(LL)W/ACCESS>CUSTOM<
- Complete these reactions. (a) (b)arrow_forwardDetermine the product formed when compound A is treated with each reagent.arrow_forwardD-Arabinose can exist in both pyranose and furanose forms.a. Draw the a and ß anomers of D-arabinofuranose.b. Draw the a and ß anomers of D-arabinopyranosearrow_forward
- Draw an example a) an aromatic heterocycle b) benzyne. c) an aromatic cation d) an aromatic anionarrow_forwardA particularly strong and rigid polyester used for electronic parts is marketed under the trade name Glyptal. It is a polymer of terephthalic acid and glycerol. Draw a segment of the polymer and explain why it is so strong.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

