
Concept explainers
(a)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(a)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
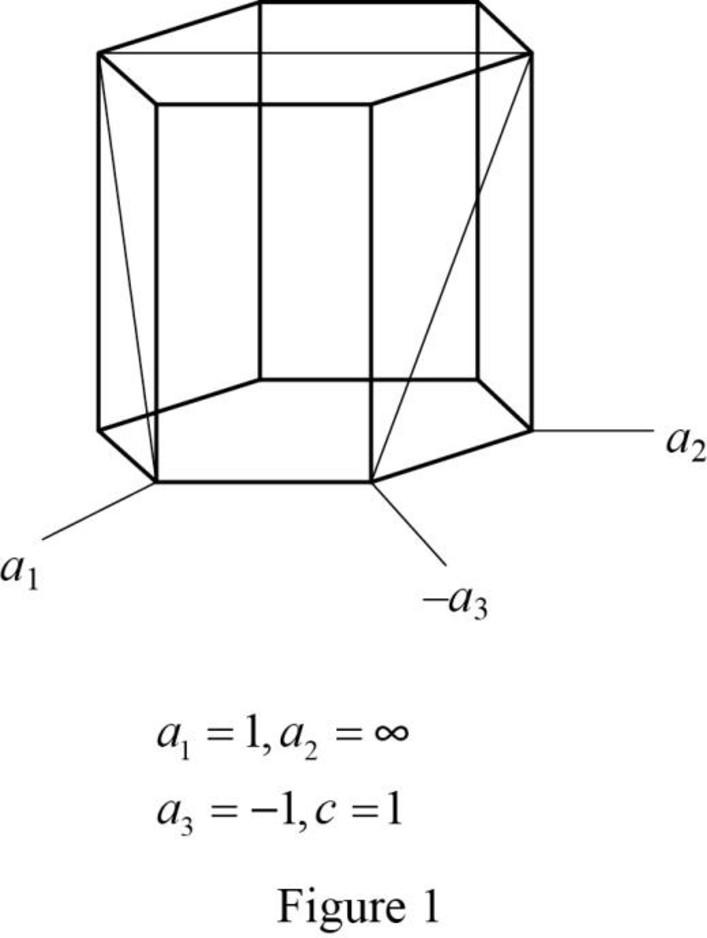
(b)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(b)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
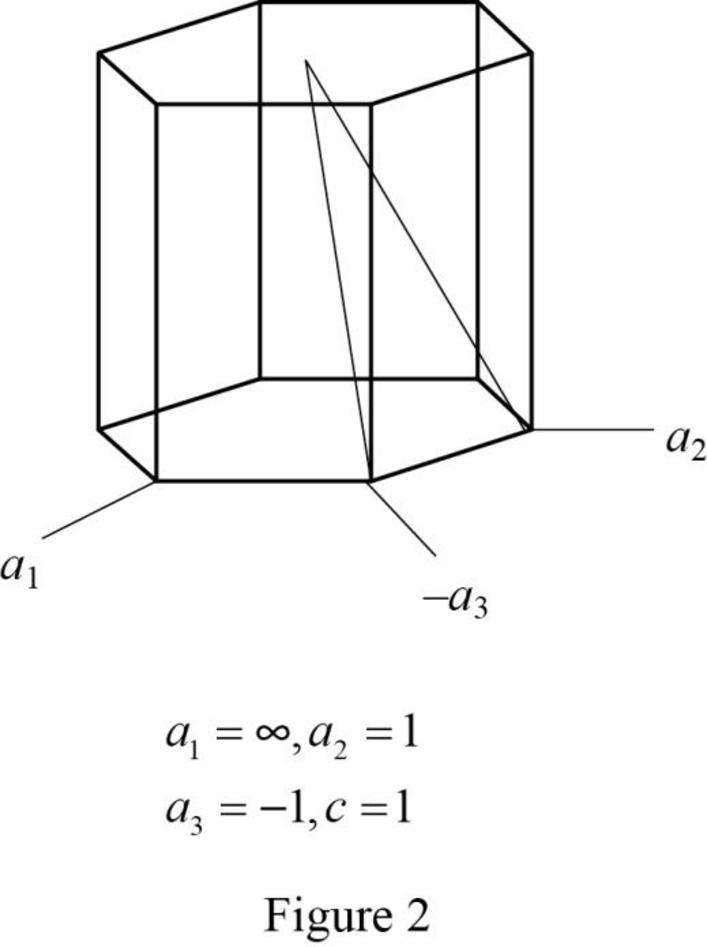
(c)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(c)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
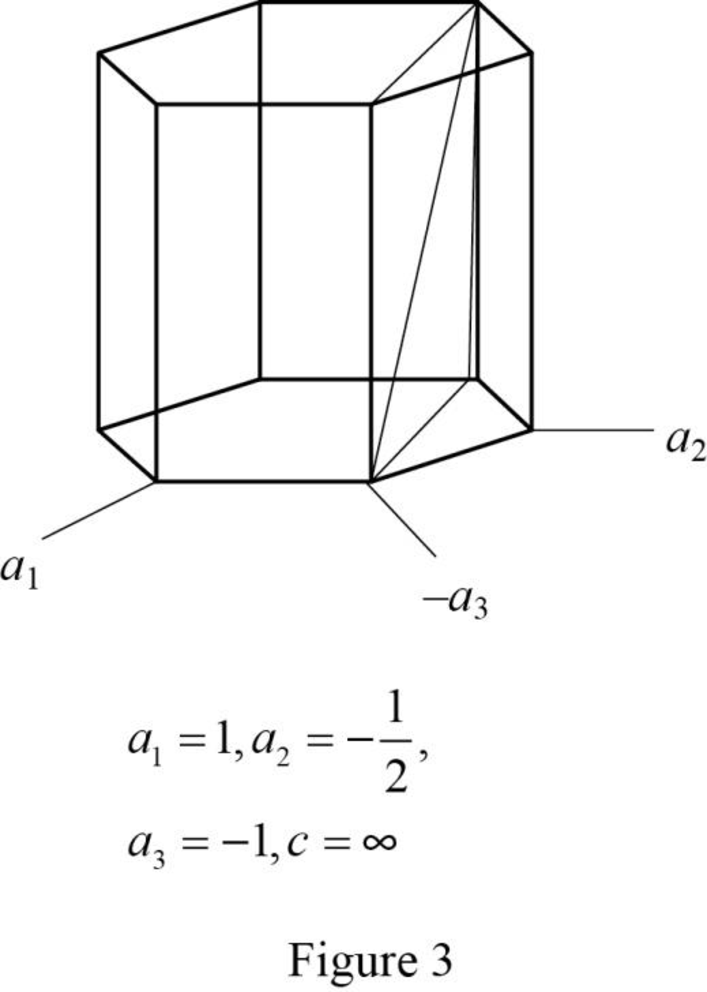
(d)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(d)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
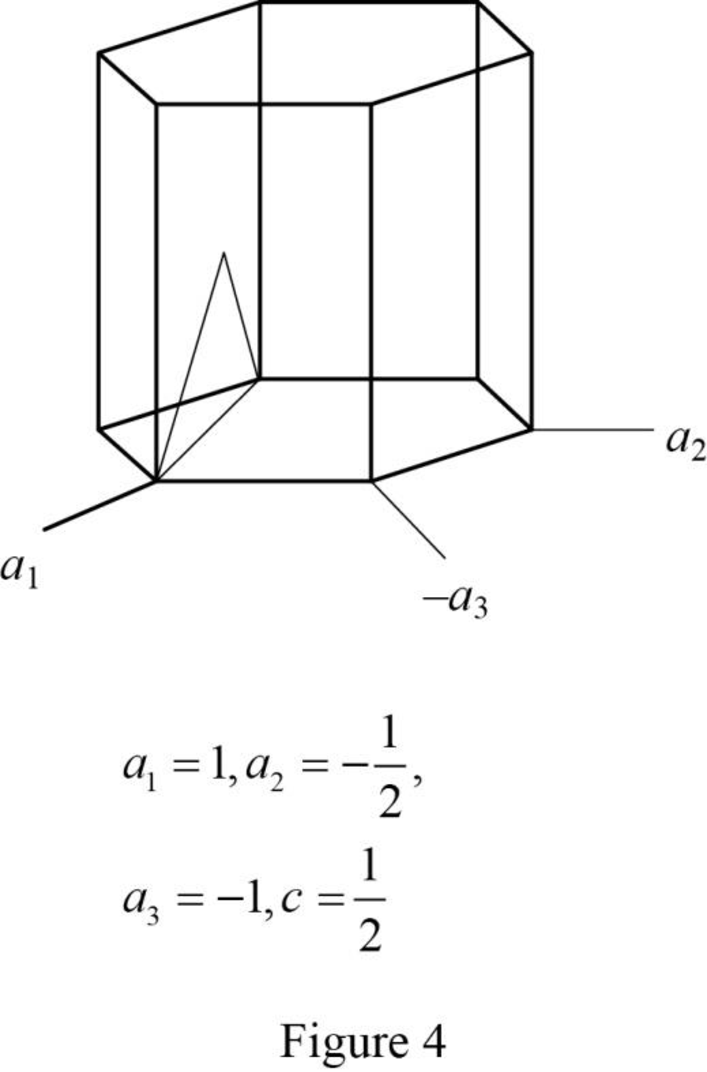
(e)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(e)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
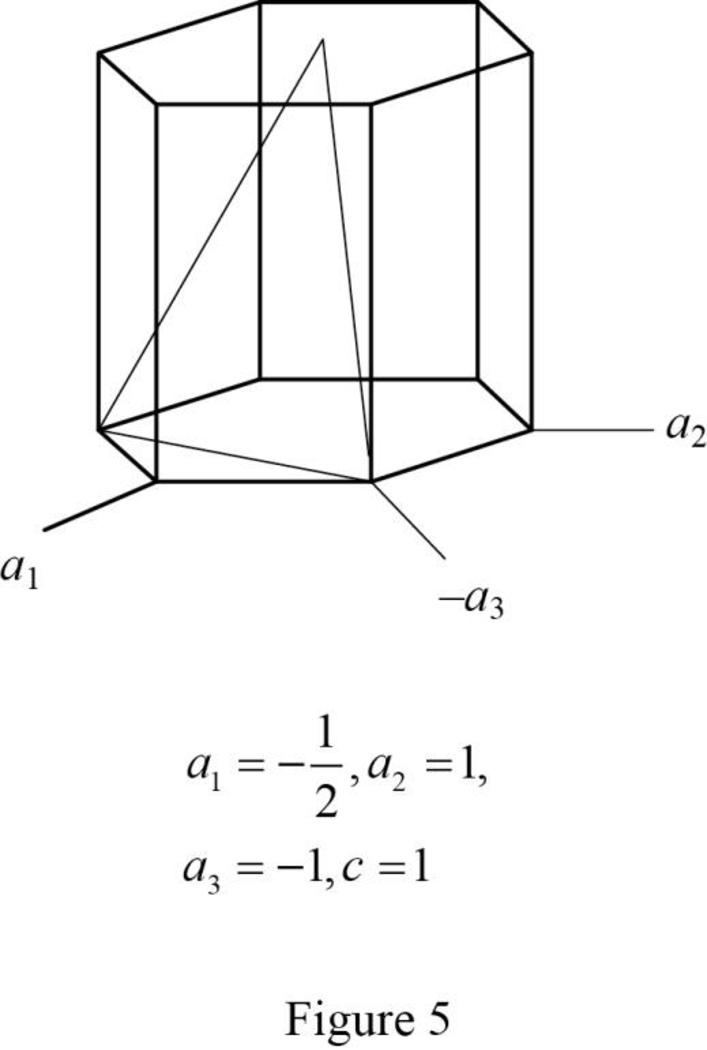
(f)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(f)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
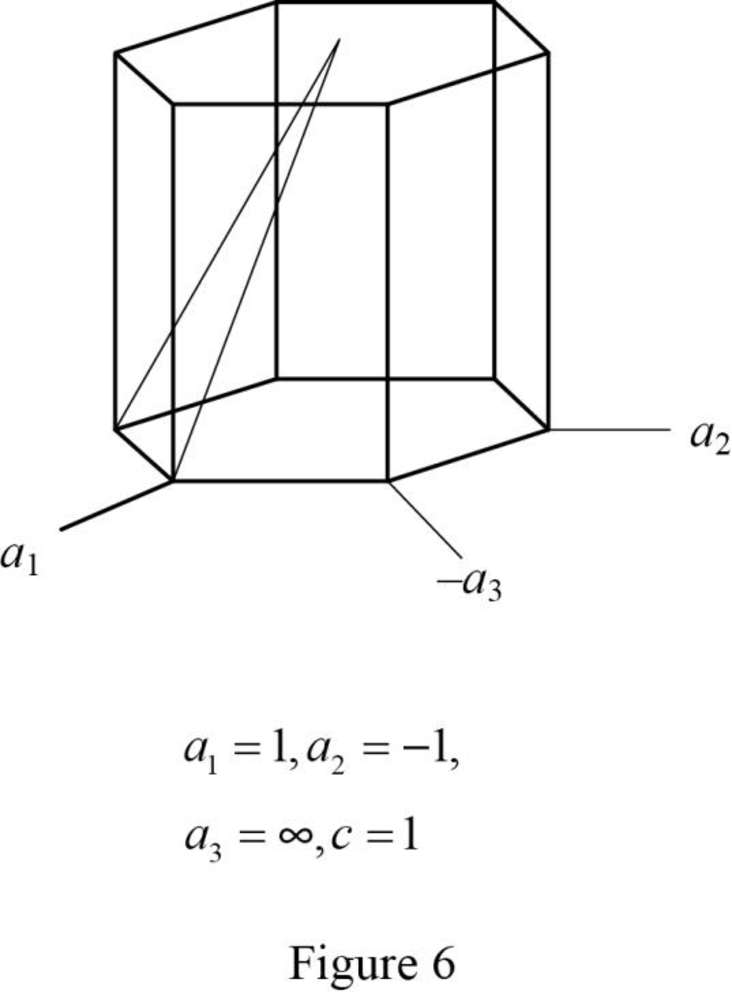
(g)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(g)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
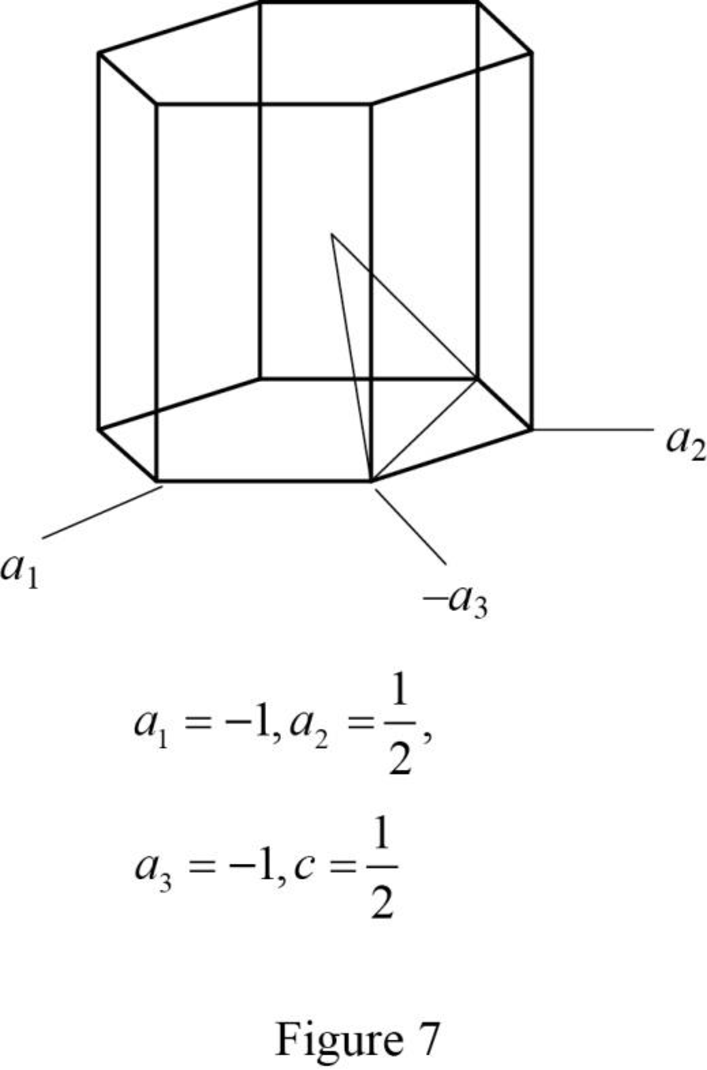
(h)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(h)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
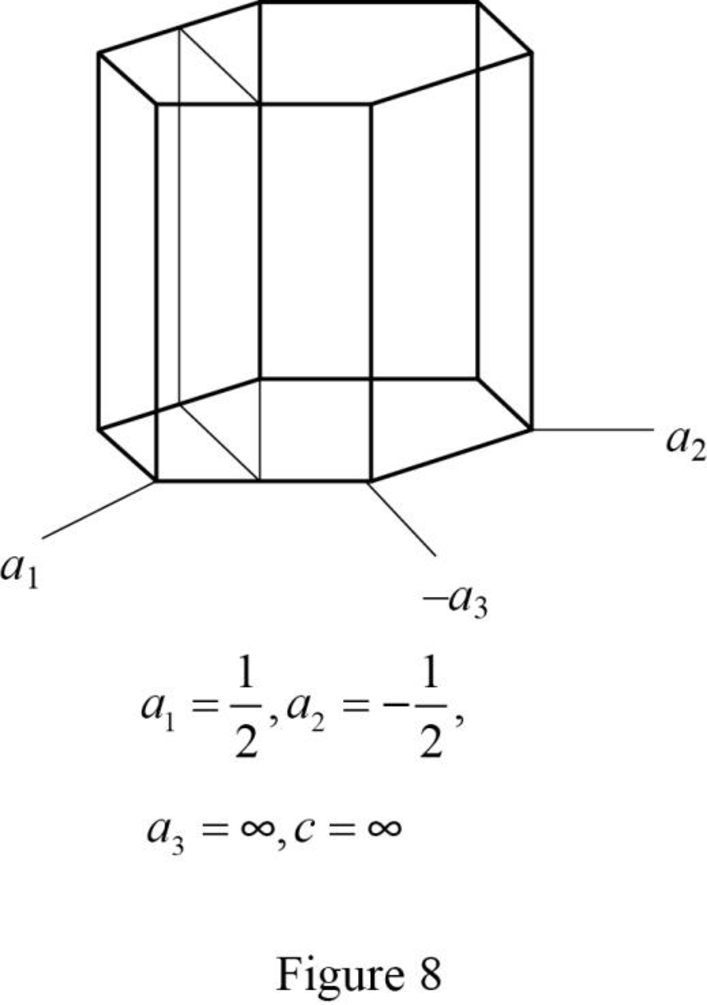
(i)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(i)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
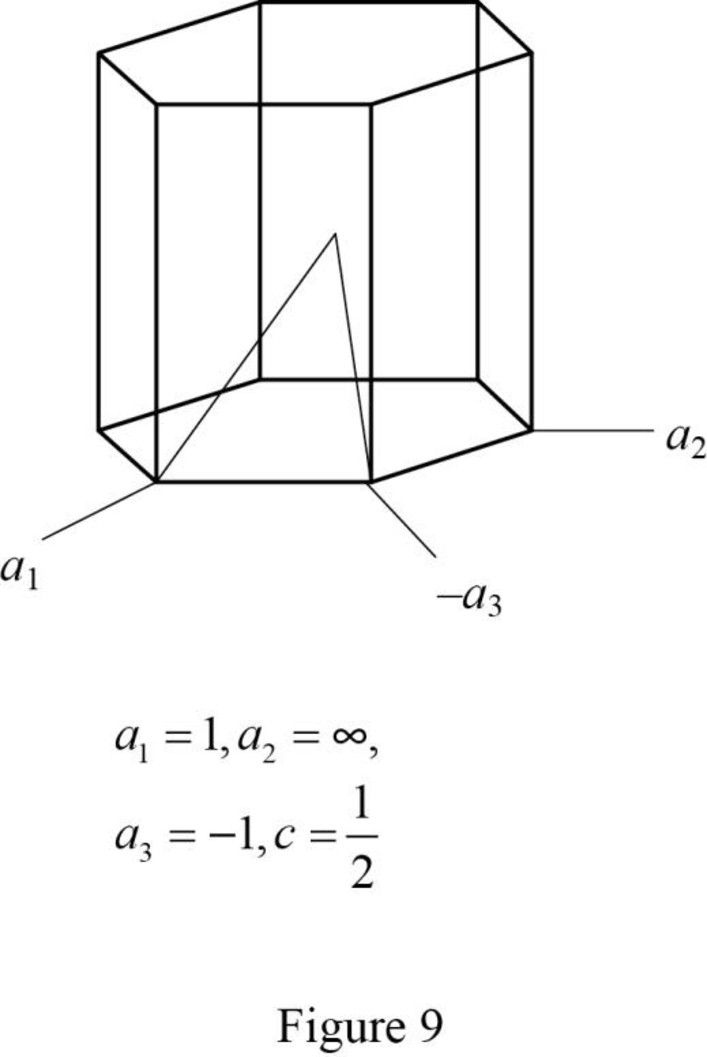
(j)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(j)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
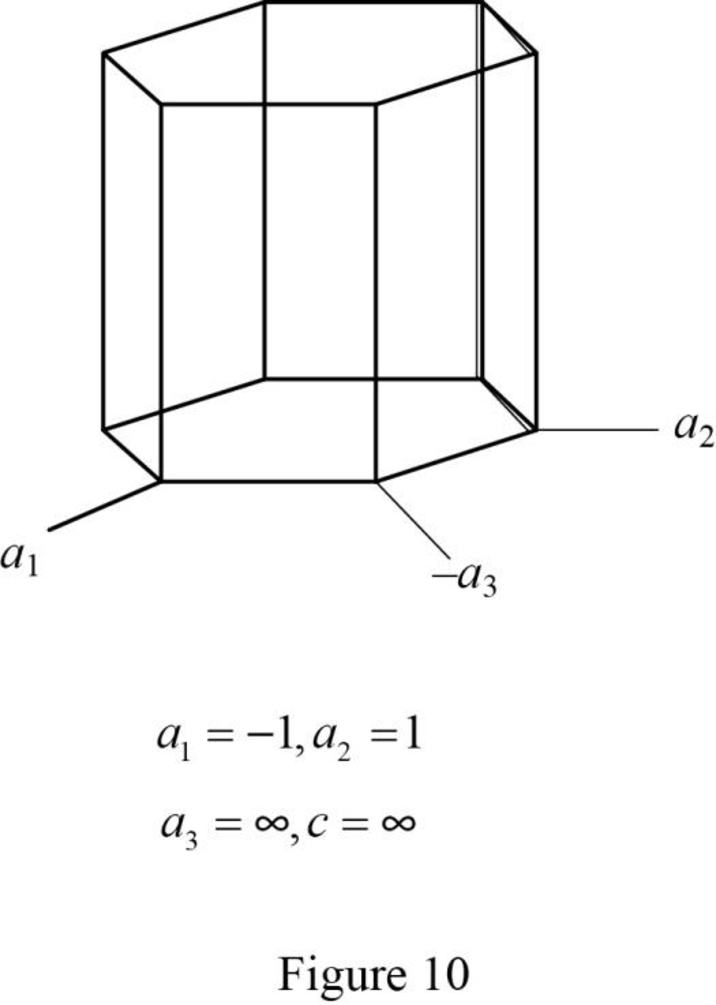
(k)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(k)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
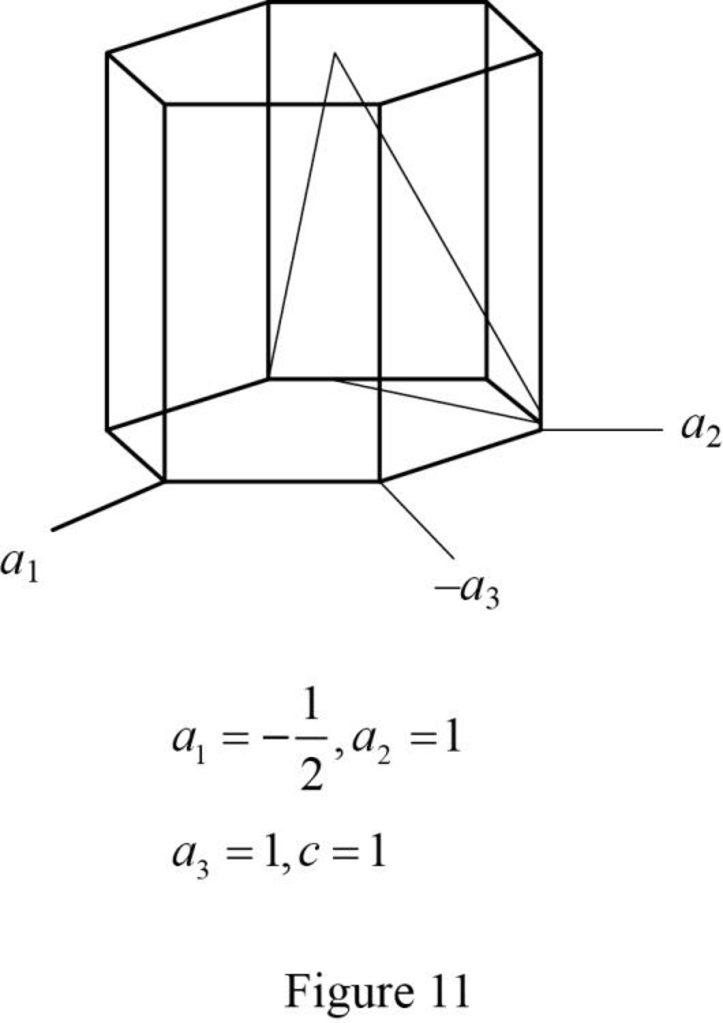
(l)
Draw the hexagonal crystal plane having Miller-Bravais indices of
(l)
Explanation of Solution
Show the hexagonal crystal plane having Miller-Bravais indices of
Want to see more full solutions like this?
Chapter 3 Solutions
FOUND.OF MTRLS.SCI+ENGR.(LL)-W/CONNECT
- For BCC metal X, compute the interplanar spacing for the 212 set of planes. The lattice parameter for Fe is 0.2194 nm. Round your answer to 2 decimal places.arrow_forwardqjly21 For FCC iron, calculate the diffraction angle for the (220) set of planes. The lattice parameter for Fe is 0.3571 nm. Assume that monochromatic radiation having a wavelength of 0.1540 nm is used, and the order of reflection is 1.arrow_forwardThe density of a sample of HCP beryllium is 1.844 g/cm 3 , and the lattice parameters are a 0 = 0.22858 nm and c 0 = 0.35842 nm. Calculate (a) the fraction of the lattice points that contains vacancies: and (b) the total number of vacancies in a cubic centimetre of Bearrow_forward
- In an ideal hexagonal closed packed system, if the atomic radius of the atom was 0.112 nm, write down the three digit value of the lattice parameter c in nm unitarrow_forwardDetermine the repeat distance, linear density, and packing fraction for FCC nickel, which has a lattice parameter of 0.35167 nm, in the[100],[110] , and [111] directions. Which of these direction is close packed?arrow_forwardoutline the procedure to determine a plane in a crystal lattice using Miller indices.arrow_forward
- a. For a FCC crystal, list all the planes in the {1 2 1} family. b. Would you expect a family in a face-centered orthorhombic (FCO) crystal to have more or fewer planes than the same family in a FCC crystal? Explain your answer.arrow_forwardNickel has the fcc crystal structure and a lattice parameter of a = 0.3517 nm. Using this information, determine the atomic radius of Ni in angstroms.arrow_forwardExplain why there is no face-centered tetragonal Bravais lattice.arrow_forward
- Calculate the radii of the largest void found in an FCC iron lattice in units of [pm] with a lattice parameter of 0.357 nm. The radius of an iron atom is approximately 0.124 nm A) 0.15 B) 10 C) 31 D) 55arrow_forwardHow many vacancies per cubic centimeter are there in gold, at ambienttemperature, assuming a lattice parameter of 0.408 nm? Gv = 1.4 × 10−19.arrow_forwardbriefly explain how with the help of x-ray diffraction techniques, the lattice dimensions are determinedarrow_forward
 Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
