
Concept explainers
Interpretation:
The
Concept Introduction:
A wave is a disturbance or variation that travels through a medium transporting energy without transporting matter. The wavelength is the distance between identical points on successive waves. The frequency is the number of waves that pass through any particular point in 1 second.
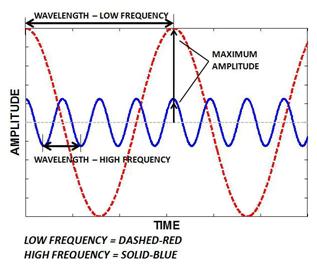
Figure1
The speed, wavelength and frequency of a wave are related by the equation:
To find: The frequency of light with wavelength of
Correct answer:
The frequency of light with wavelength of
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Student Solutions Manual For Chemistry: Atoms First
- If photons have a frequency of 1.269x1015 s-1, what wavelength, in nm, does this correspond to?answer in sig figsarrow_forwardThe energy for one state of the H atom is -5.45 × 10 -19 J. What frequency (Hz) of light would need to be absorbed to reach a state with an energy of -8.72 × 10 -20 J? Express answer in scientific notation.arrow_forwardWhat is the wavelength of light (nm) that has a frequency of 3.22 × 1014 s-1?arrow_forward
- A particular transition of the rubidium atom emits light whose frequency is 3.84 1014 Hz. (Hz is the abbreviation for hertz, which is equivalent to the unit/s, or s1.) Is this light in the visible spectrum? If so, what is the color of the light? (See Figure 7.5.)arrow_forwardWhich of the following are applicable when explaining the photoelectric effect? Correct any statements that are wrong. (a) Light is electromagnetic radiation. (b) The intensity of a light beam is related to its frequency. (c) Light can be thought of as consisting of mass- less particles whose energy is given by Plancks equation, E = h.arrow_forwardH2. 2. Calculate the energy of transition from n = 3 to n = 1. In what region of electromagnetic spectrum is this radiation found? Please give typed answerarrow_forward
- What is the wavelength (in nm) of a photon required to excite an electron from n=2 to n= 5 in a He+ ion?. Answer to three significant figuresarrow_forwardA radio wave has a frequency of 2.80 ×108 Hz. What is the energy (in J) of one photon of this radiation? Answer in scientific notation.arrow_forwardCalculate the wavelength of light emitted from a hydrogen atom when it undergoes a transition from the n=8 level to n=5.*Enter the correct numeric value to 2.0% precision. Express scientific notation like 4.29E-15arrow_forward
- Calculate the photon energy associated with a transition from the n = 5 level to the n = 3 level in a Be3+ Is a photon being absorbed or emitted in this transition? With any calculation, would you expect the photon energy to be larger or smaller for the transition from the n = 5 level to the n = 4 level? (b and c can be 1 word answers)arrow_forwardWhat is the frequency of a photon resulting from the transition of n = 6 → n = 1 ? (give answer to 3 digits)arrow_forward1)Which statements are incorrect? (1) UV light has a lower frequency than Visible light.(2) UV light has a lower wavelength than Visible light.(3) The speed of IR light is directly proportional to its wavelength and inversely proportional to its frequency. Group of answer choices only (1) OR only (2) OR only (3) (1) AND (2) AND (3) (1) and (3) (1) and (2) (2) and (3)arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning  Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



