
Interpretation:
The waves having the greatest frequency, the greatest wavelength and the greatest amplitude should be described using the relation between speed, wavelength and frequency of a wave.
Concept Introduction:
A wave is a disturbance or variation that travels through a medium transporting energy without transporting matter. The wavelength is the distance between identical points on successive waves. The frequency is the number of waves that pass through any particular point in 1 second.
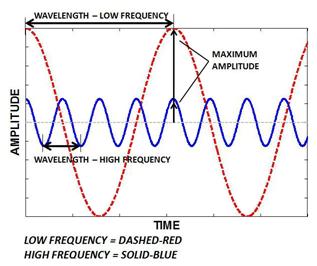
Figure1
The speed, wavelength and frequency of a wave are related by the equation:
To Describe: The waves having the greatest frequency, the greatest wavelength and the greatest amplitude
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Student Solutions Manual For Chemistry: Atoms First
- What is the energy in Joules of one mole of photons associated with radiation that has frequency of 3.41 x 10 ^15 Hz? Use 1 decimal place in answer please. Thanks :)arrow_forwardWhat is the wavelength (in m) of a 98.4 MHz signalarrow_forwardWhat is the wavelength of radiation that has a frequency of 2.10 x 1014 s-1? (Answer in meters, to 3 sig figs.arrow_forward
- What is the wavelength of a photon of red light (in nm) whose frequency is 4.60 x 1014 Hz? Group of answer choices 652 nm 153 x 106 nm 153 nm 460 nmarrow_forward337.1 nm(wavelength of a nitrogen laser) Express your answer using three significant figures.arrow_forwardA particular transition of the rubidium atom emits light whose frequency is 3.84 1014 Hz. (Hz is the abbreviation for hertz, which is equivalent to the unit/s, or s1.) Is this light in the visible spectrum? If so, what is the color of the light? (See Figure 7.5.)arrow_forward
- At its closest approach, Mercury is 77.3 million km from Earth. How long would it take to send a radio message from a space probe of Mercury to Earth when the planets are at this closest distance?arrow_forwardWhat is the wavelength, in nm, of an H2 molecule (3.32 x10^-27 kg) traveling at 2.75 x 10^3 m/s? Use 4 decimal places in answer please. Thanks.arrow_forwardSuppose you are standing 225 m from a radio transmitter. What is your distance from the transmitter in terms of the number of wavelengths if(a) the station is broadcasting at 1150 kHz (on the AM radio band)? (1kHz = 1 × 10 3Hz) (b) the station is broadcasting at 98.1 MHz (on the FM radio band)? (1 MHz × 10 6arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning  Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





