
Concept explainers
(a)
The sketch of wave function.
(a)
Explanation of Solution
Introduction:
The wave function contains all information about a system. The graph of wave function for state
For
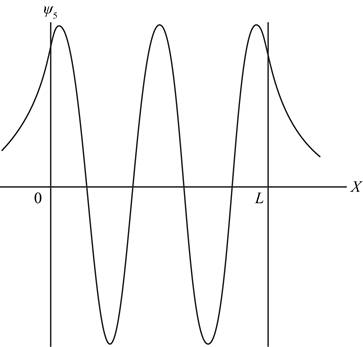
Figure 1
Conclusion:
Therefore, the graph of wave function has five extrema as shown in figure 1.
(b)
The sketch of probability density function.
(b)
Explanation of Solution
Introduction:
The probability density function is the measure of the area under the curve of density function between the given intervals above the horizontal axis.
For
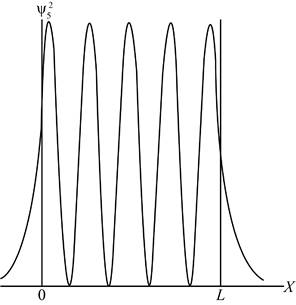
Figure 2
Conclusion:
Therefore, the graph of probability density function is shown in figure 2.
Want to see more full solutions like this?
Chapter 35 Solutions
Physics For Scientists And Engineers Student Solutions Manual, Vol. 1
- Find the expectation value x2 of the square of the position for a quantum harmonic oscillator in the ground state. Note: +dxx2ea x 2=(2a 3/2)1.arrow_forwardAn electron is trapped in a finite potential well that is deep enough to allow the electron to exist in a state with n= 4. How many points of (a) zero probability and (b) maximum probability does its matter wave have within the well?arrow_forwardThe ground-state energy of an electron trapped in a onedimensional infinite potential well is 2.6 eV.What will this quantity be if the width of the potential well is doubled?arrow_forward
- It is true that the particles in a one-dimensional potential well can exist only in states of definite energy described by the wave functions? These states are labeled by the quantum number n.arrow_forwardWhat is the ground-state energy of (a) an electron and (b) a proton if each is trapped in a one-dimensional infinite potential well that is 200 pm wide?arrow_forwardAn electron is trapped in a one-dimensional infinite potential well that is 460 pm wide; the electron is in its ground state. What is the probability that you can detect the electron in an interval of width δx = 5.0 pm centered at x = 300 pm? (Hint: The interval δx is so narrow that you can take the probability density to be constant within it.)arrow_forward
- An electron is trapped in a one-dimensional infinite potential well that is 100 pm wide; the electron is in its ground state. What is the probability that you can detect the electron in an interval of width x = 5.0 pm centered at x = (a) 25 pm, (b) 50 pm, and (c) 90 pm? (Hint: The interval x is so narrow that you can take the probability density to be constant within it.)arrow_forwardA particle of mass m mves in the infinite square well potential v(x)= 0. if -a/2 to a/2 infinitive find <p> and <p^2> using wave function shai 1 and compute <p> and <p^2>arrow_forwardAn electron, trapped in a one-dimensional infinite potential well 250 pm wide, is in its ground state. How much energy must it absorb if it is to jump up to the state with n= 4?arrow_forward
- An electron is trapped in a one-dimensional infinite potential well. For what (a) higher quantum number and (b) lower quantum number is the corresponding energy difference equal to the energy of the n= 5 level? (c) Show that no pair of adjacent levels has an energy difference equal to the energy of the n = 6 level.arrow_forwardSuppose a wave function is discontinuous at some point. Can this function represent a quantum state of some physical particle? Why? Why not?arrow_forwardA particle of mass m is confined to a box of width L. If the particle is in the first excited state, what are the probabilities of finding the particle in a region of width0.020 L around the given point x: (a) x=0.25L; (b) x=040L; (c) 0.75L and (d) x=0.90L.arrow_forward
 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning



