
Concept explainers
Flow of Water from a Tank Water flows from a tank of constant cross-sectional area 50 ft2 through an orifice of constant cross-sectional area 1.4 ft2 located at the bottom of the tank (see the figure).
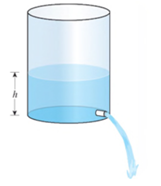
Initially, the height of the water in the tank was 20 ft, and its height t sec later is given by the equation
How fast was the height of the water decreasing when its height was 8 ft?
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Applied Calculus for the Managerial, Life, and Social Sciences: A Brief Approach
- Radius of a Shock Wave An explosion produces a spherical shock wave whose radius R expands rapidly. The rate of expansion depends on the energy E of the explosion and the elapsed time t since the explosion. For many explosions, the relation is approximated closely by R=4.16E0.2t0.4. Here R is the radius in centimeters, E is the energy in ergs, and t is the elapsed time in seconds. The relation is valid only for very brief periods of time, perhaps a second or so in duration. a. An explosion of 50 pounds of TNT produces an energy of about 1015 ergs. See Figure 2.71. How long is required for the shock wave to reach a point 40 meters 4000 centimeters away? b. A nuclear explosion releases much more energy than conventional explosions. A small nuclear device of yield 1 kiloton releases approximately 91020 ergs. How long would it take for the shock wave from such an explosion to reach a point 40 meters away? c. The shock wave from a certain explosion reaches a point 50 meters away in 1.2 seconds. How much energy was released by the explosion? The values of E in parts a and b may help you set an appropriate window. Note: In 1947, the government released film of the first nuclear explosion in 1945, but the yield of the explosion remained classified. Sir Geoffrey Taylor used the film to determine the rate of expansion of the shock wave and so was able to publish a scientific paper concluding correctly that the yield was in the 20-kiloton range.arrow_forwardVolume of Grain Grain is falling from a chute onto the ground, forming a conical pile whose diameter is always three times its height. How high is the pile (to the nearest hundredth of a foot) when it contains 1000 ft3 of grain?arrow_forwardVan der Waals Equation In Exercise 18 at the end of Section 2.3, we discussed the ideal gas law, which shows the relationship among volume V, pressure p, and temperature T for a fixed amount 1 mole of a gas. But chemists believe that in many situations, the van der Waals equation gives more accurate results. If we measure temperature T in kelvins, volume V in liters, and pressure p in atmosphere 1 atm is the pressure exerted by the atmosphere at sea level, then the relationship for carbon dioxide is given by p=0.082TV0.0433.592V2atm What volume does this equation predict for 1 mole of carbon dioxide at 500 kelvins and 100 atm?Suggestion: Consider volumes ranging from 0.1 to 1 liter.arrow_forward
- Air Temperature As dry air moves upward, it expand and, in so doing, cools at a rate of about 1°C for each 100-meter rise, up to about 12 km. (a) If the ground temperature is 20°C, write a formula for the temperature at height h. (b) What range of temperatures can be expected if an air plane lakes off and reaches a maximum height of 5 km?arrow_forwardThe Ideal Gas Law A mole of a chemical is a fixed number, like a dozen, of molecules or atoms in the case of an element of that compound. A mole of water, for example, is about 18 grams, or just over half an ounce in your kitchen. Chemists often use the mole as the measure of the amount of a chemical compound. A mole of carbon dioxide has a fixed mass, but the volume V that it occupies depends on pressure p and temperature T; greater pressure tends to compress the gas into a smaller volume, whereas increasing temperature tends to make the gas expand into a larger volume. If we measure the pressure in atmospheres 1 atm is the pressure exerted by the atmosphere at sea level, the temperature in kelvins, and the volume in liters, then the relationship is given by the ideal gas law: pV=0.082T. a. Solve the ideal gas law for the volume V. b. What is the volume of 1 mole of carbon dioxide under 3 atm of pressure at a temperature of 300 kelvins? c. Solve the ideal gas law for pressure. d. What is the pressure on 1 mole of carbon dioxide if it occupies a volume of 0.4 liter at a temperature of 350 kelvins? e. Solve the ideal gas law for temperature. f. At what temperature will 1 mole of carbon dioxide occupy a volume of 2 liters under a pressure of 0.3 atm?arrow_forwardThe displacement from equilibrium of an oscillating weight suspended by a spring is given by y(t)=3sin(t/4), where y is the displacement in feet and t is the time in seconds. Complete the table Use the table feature of a graphing utility to determine when the displacement is maximum. Use the table feature of the graphing utility to approximate the time t (0 < t < 8) when the weight reaches equilibrium.arrow_forward
 College AlgebraAlgebraISBN:9781305115545Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage LearningAlgebra & Trigonometry with Analytic GeometryAlgebraISBN:9781133382119Author:SwokowskiPublisher:Cengage
College AlgebraAlgebraISBN:9781305115545Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage LearningAlgebra & Trigonometry with Analytic GeometryAlgebraISBN:9781133382119Author:SwokowskiPublisher:Cengage Algebra and Trigonometry (MindTap Course List)AlgebraISBN:9781305071742Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage Learning
Algebra and Trigonometry (MindTap Course List)AlgebraISBN:9781305071742Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage Learning Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning
Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning Trigonometry (MindTap Course List)TrigonometryISBN:9781337278461Author:Ron LarsonPublisher:Cengage Learning
Trigonometry (MindTap Course List)TrigonometryISBN:9781337278461Author:Ron LarsonPublisher:Cengage Learning




