
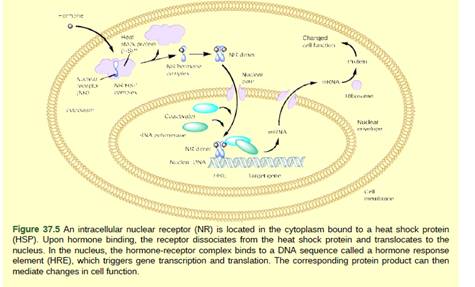
Figure 37.5 Heat shock proteins (HSP) are so named because they help refold misfolded proteins. In response to increased temperature (a “heat shock"), heat shock proteins are activated by release from the NR/HSP complex. At the same time, transcription of HSP genes is activated. Why do you think the cell responds to a heat shock by increasing the activity of proteins that help refold misfolded proteins?
To write:
The reasons behind the cell response to a heat shock to increase the activity of proteins that help refold misfolded proteins.
Introduction:
Heat shock proteins are the proteins, which act as the molecular chaperons. These are only produced in the cells when the cells undergo the stressed conditions. They play the functions in stabilizing the proteins and ensure the folding and refolding of the proteins.
Explanation of Solution
HSPs regulate the process of transcription. When the cell faced any stressed conditions, the intracellular nuclear receptor bond to these heat shock proteins and gets dissociated from the heat shock proteins upon hormone binding. This nuclear receptor and heat shock protein complexes activate the heat shock proteins and the process of transcription and translation take place.
After this, the hormone receptor complex binds a DNA sequence in the nucleus and form hormone response element. This element functions in initiating the process of gene transcription and translation and the resulting corresponding protein product will regulate the changes functions of cells. In case of high temperature, the structure of the protein gets unfold or get denature.
In case of high temperature, the proteins structure get unfold or get denature.
Want to see more full solutions like this?
Chapter 37 Solutions
BIOLOGY
Additional Science Textbook Solutions
College Physics
Microbiology with Diseases by Body System (4th Edition)
Campbell Biology: Concepts & Connections (9th Edition)
Campbell Essential Biology (6th Edition) - standalone book
Human Biology: Concepts and Current Issues (8th Edition)
Biological Science (6th Edition)
- What triggers the activation of G proteins? When triggered, what do G proteins do? Explain how a signal transduction process within a cell may be inactivated.arrow_forwardWhy is the GTPase activity of G proteins crucial to the proper functioning of a cell? Why have G proteins not evolved to catalyze GTP hydrolysis more efficiently?arrow_forwardWhy is the GTPase activity of G proteins crucial to the proper functioning of a cell? Propose a theory as to why G proteins have not evolved to catalyze GTP hydrolysis more efficiently.arrow_forward
- In order to metabolize ethanol, the liver uses a pathway that involves alcohol dehydrogenase and aldehyde dehydrogenase. Both of these enzymes generate NADH,NADH, which then accumulates, contributing to the development of fatty liver disease. SELECT ALL of the processes that describe the effects of NADHNADH accumulation? a.promotion of triacylglycerol hydrolysis b.promotion of fatty acid synthesis c.inactivation of transcription factor d.changes in gene transcriptionarrow_forwardUnder what conditions does the CAP protein become a functional activating protein? Group of answer choices when cAMP is absent and CAP is bound to glucose when glucose is absent and CAP is bound to cAMP none of these when glucose is abundant and CAP is bound to cAMP when cAMP is abundant and CAP is bound to glucosearrow_forwardA drug that inhibits the kinase activity of AKT would MOST likely have which of the following effects? A. Increased GSK-3b activity and increased CyclinD synthesis B. Increased GSK-3b activity and decreased CyclinD synthesis C. Decreased GSK-3b activity and increased CyclinD synthesis D. Decrease GSK-3b activity and decreased CyclinD synthesisarrow_forward
- Cholera toxins, produced by the pathogenic bacteria Vibrio cholerae, disrupt G Protein Coupled Receptor (GPCR) signaling pathways. They interfere with... the ability of the beta subunit of a G protein to bind to the GPCR the ability of the gamma subunit of a G protein to exchange GDP for GTP the ability of the beta subunit of a G protein to bind to an effector protein the ability of the alpha subunit of a G protein to hydrolyze GTP to GDParrow_forwardWhich of the following signaling mechanisms is not a switch mechanism? a. Changes in phosphorylation state b. Binding of GTP c. Binding of Ca2+ ion d. Protease activityarrow_forwardTSC acts as a tumor suppressor by continually suppressing mTOR and must be inhibited in order for mTOR to be active. True or false, explain whyarrow_forward
- The cell is triggered to make insulin when a glucose molecule binds to a receptor on the surface of the beta-cell. This then activates a molecule inside the cell to activate the expression of the gene. This process in which cells communicate with each other is called ___________ pathway (two words).arrow_forwardThe Iron Response Factor (IRF) protein regulates production of both the iron transport protein transferrin, which imports iron into a cell, and the iron storage protein ferritin, which stores iron when the cell has more iron than in needs. IRF is an RNA binding protein, which binds to a specific RNA sequence, the Iron Response Element (IRE). When the IRF protein binds to the IRE sequence in the mRNA that codes for transferrin, what effect does this have on the expression of transferrin? Briefly explain why this effect is observed.arrow_forwardThe lipid-soluble thyroxine hormone binds a nuclear receptor that is also a transcription factor. Binding of the thyroxine hormone will therefore result in A change in the activity of different proteins in the cytoplasm A change in the expression of specific genes An activation of a second messenger pathwayarrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
