
Concept explainers
Carbon dioxide gas enters a pipe at 3 MPa and 500 K at a rate of 2 kg/s. CO2 is cooled at constant pressure as it flows in the pipe, and the temperature of the CO2 drops to 450 K at the exit. Determine the volume flow rate and the density of carbon dioxide at the inlet and the volume flow rate at the exit of the pipe using (a) the ideal-gas equation and (b) the generalized compressibility chart. Also, determine (c) the error involved in the first case.
FIGURE P3–89
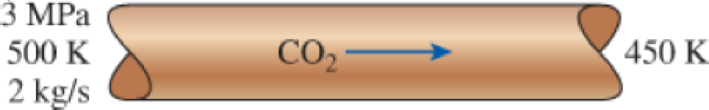
(a)
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the ideal gas equation of state.
Answer to Problem 89P
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the ideal gas equation of state are
Explanation of Solution
Refer to Table A-1, obtain the gas constant, critical pressure, and the critical temperature of carbon dioxide.
Write the equation of volume flow rate at the inlet of the pipe.
Here, inlet temperature and inlet pressure are
Calculate the density at the inlet of pipe.
Calculate the equation of volume flow rate at the outlet of the pipe.
Here, outlet temperature and outlet pressure are
Conclusion:
Substitute
Substitute
Substitute
Thus, the volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the ideal gas equation of state are
(b)
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the generalized compressibility chart.
Answer to Problem 89P
The volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the generalized compressibility chart are
Explanation of Solution
Calculate the equation of reduced pressure at the inlet of the pipe.
Here, the critical pressure is
Calculate the equation of reduced temperature at the inlet of the pipe.
Here, the critical temperature is
Calculate the equation of reduced pressure at the outlet of the pipe.
Calculate the equation of reduced temperature at the outlet of the pipe.
Write the equation of volume flow rate at the inlet of the pipe.
Here, compressibility factor at the inlet of pipe is
Calculate the density at the inlet of pipe.
Calculate the equation of volume flow rate at the outlet of the pipe.
Here, compressibility factor at the outlet of pipe is
Conclusion:
Substitute 3 MPa for
Substitute 500 K for
Substitute 3 MPa for
Substitute 450 K for
Refer to Figure 3-48, obtain the compressibility factor at inlet state
Refer to Figure 3-48, obtain the compressibility factor at outlet state
Substitute 0.9791 for
Substitute 0.9791 for
Substitute 0.9656 for
Thus, the volume flow rate, density of carbon dioxide at the inlet, and the volume flow rate at the exit of the pipe using the generalized compressibility chart are
(c)
The error involved in the first case.
Answer to Problem 89P
The error involved in the first case are
Explanation of Solution
Calculate the percentage of error involved in the first case of volume flow rate at the inlet condition.
Here, calculated volume flow rate at inlet state from EOS is
Calculate the percentage of error involved in the first case of density at the inlet condition.
Here, calculated density at inlet state from EOS is
Calculate the percentage of error involved in the first case of volume flow rate at the outlet condition.
Here, calculated volume flow rate at outlet state from EOS is
Conclusion:
Substitute
Substitute
Substitute
Thus, the error involved in the first case are
Want to see more full solutions like this?
Chapter 3 Solutions
Thermodynamics: An Engineering Approach
- Refrigerant-134a at 400 psia has a specific volume of 0.1384 ft3 /lbm. Determine the temperature of the refrigerant based on (a) the ideal-gas equation, (b) the generalized compressibility chart, and (c) the refrigerant tables.arrow_forwardThe piston in a car engine compresses a mixture of gasoline vapour and air from 700ml to 100ml during its compression stroke. Before the compression stroke, the mixture had a pressure of 75 kPa and a temperature of 120celcius. What is the pressure of the cylinder after the compression stroke if the temperature of the gas rises to 200celcius? Express your answer in MPa with two significant digits.arrow_forwardWater is boiled at sea level in a coffeemaker equipped with an immersion-type electric heating element. The coffeemaker contains 1 L of water when full. Once boiling starts, it is observed that half of the water in the coffeemaker evaporates in 13 min. Determine the power rating of the electric heating element immersed in water. Also, determine how long it will take for this heater to raise the temperature of 1 L of cold water from 18°C to the boiling temperature.arrow_forward
- Determine the specific volume, in m3/kg, of R-134a at a pressure of 100 kPa and temperature of 10°C.arrow_forwardA 9-m3 tank contains nitrogen at 17°C and 600 kPa. Some nitrogen is allowed to escape until the pressure in the tank drops to 400 kPa. If the temperature at this point is 15°C, determine the amount of nitrogen that has escaped.arrow_forwardA 5-ft3 rigid tank contains a saturated mixture of refrigerant-134a at 50 psia. If the saturated liquid occupies 20 percent of the volume, determine the quality and the total mass of the refrigerant in the tank.arrow_forward
- What is the percentage of error involved in treating carbon dioxide at 5 MPa and 25°C as an ideal gas?arrow_forwardDraw a P-V Diagram for a gas that goes through a two-step process. (1) The first step is isobaric; (2) and the second step is isochoric.arrow_forwardA 1-kg steam-water mixture at 1.0 MPa is contained in an inflexible tank. Heat is added until the pressure rises to 3.5 MPa and the temperature to 400°C. Determine the heat added in kJ.arrow_forward
- Steam at a pressure of 3.5 MPa is known to have a specific volume of 50x10^-3 m^3/kg. What is its specific enthalpy?arrow_forwardwhy water evaporates at constant pressure at constant temperature, and how the temperature is when the pressure is different?arrow_forward0.05 kg of steam at 1.5 MPa is contained in a rigid vessel of volume of 0.0076 m^3. A. What is the temperature of the steam? B. If the vessel is cooled, at what temperature will the steam be just dry and saturated? C. Cooling is continued until the pressure in the vessel is 1.1 MPa, calculate the amount of vapor at this point.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





