
Concept explainers
a)
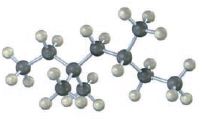
Interpretation:
The skeletal structures of the following compounds are to be drawn, thereby determining the IUPAC name of them.
Concept introduction:
The skeletal structures of the following compounds are drawn and the following IUPAC name can be given.
1. Parent chain (Root word)
2. Prefix (substituents)
3. Suffix (depends on the parent chain)
b)
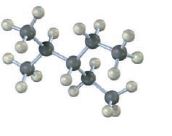
Interpretation:
The skeletal structures of the following compounds are to be drawn, thereby determining the IUPAC name of them.
Concept introduction:
The skeletal structures of the following compounds are drawn and the following IUPAC name can be given.
1. Parent chain (Root word)
2. Prefix (substituents)
3. Suffix (depends on the parent chain)
c)
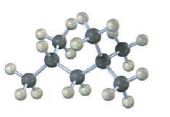
Interpretation:
The skeletal structures of the following compounds are to be drawn, thereby determining the IUPAC name of them.
Concept introduction:
The skeletal structures of the following compounds are drawn and the following IUPAC name can be given.
1. Parent chain (Root word)
2. Prefix (substituents)
3. Suffix (depends on the parent chain)
d)
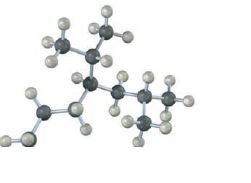
Interpretation:
The skeletal structures of the following compounds are to be drawn, thereby determining the IUPAC name of them.
Concept introduction:
The skeletal structures of the following compounds are drawn and the following IUPAC name can be given.
1. Parent chain (Root word)
2. Prefix (substituents)
3. Suffix (depends on the parent chain)
Trending nowThis is a popular solution!

Chapter 3 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- draw structural formulas for all isomeric alkanes with molecular formula C7H16. Predict which isomer has the lowest boiling point and which has the highest boiling point.arrow_forwardCompounds A and B are isomers of the molecular formula C9H19Br. Both yield the same alkene C in an elimination reaction. Hydrogenation of C yields the product 2,3,3,4 tetramethyl pentane. What are the structures of A, B, and C?arrow_forwardWrite condensed and skeletal structures for all the tertiary alcohols with molecular formula C6H14O and give each a systematic name.arrow_forward
- Propose a structural formula for the product(s) when each of the following alkenes is treated with H2O/H,SO4. Why are two products formed in part (b), but only one in parts (a) and (c)? (a) 1-Hexene gives one alcohol with a molecular for- mula of C,H140. (b) 2-Hexene gives two alcohols, each with a molecu- lar formula of C,H140. (c) 3-Hexene gives one alcohol with a molecular for- mula of C6H140.arrow_forwardThe melting points and boiling points of two isomeric alkanes are as follows: CH3(CH2)6CH3, mp = -57 °C and bp = 126 °C; (CH3)3CC(CH3)3, mp = 102 °C and bp = 106 °C. (a) Explain why one isomer has a lower melting point but higher boiling point. (b) Explain why there is a small difference in the boiling points of the two compounds, but a huge difference in their melting points.arrow_forwardOne mole of an unknown hydrocarbon, compound C, in the presence of a platinum catalyst, adds 98.9 L of hydrogen, measured at 744 mm Hg and 22 degrees C , to form a saturated alkane which contains one ring. When one mole of compound C is reacted with ozone, followed by reduction with (CH3)2S , four moles of only one product was formed, whose condensed molecular formula is CHO -CHO. Give the structure of compound C. Explain your reasoningarrow_forward
- What alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?arrow_forwardBased on naming branched alkyl substituents, and draw all possible alkyl groups having the formula C5H11–. Give the IUPAC names for the eight compounds of molecular formula C10H20that contain a cyclopentane ring with each of these alkyl groups as a substituent.arrow_forwardName and draw structural formulas for all cycloalkanes with molecular formula C5H10. Include cis and trans isomers as well as constitutional isomers.arrow_forward
- Give the systematic names for all alkanes with molecular formula C7H16 that do not have any secondary hydrogens.arrow_forwardAn alkene having 7 carbon atoms has Molecular formula and its name is O C;H14, heptene O C7H14, hexene C7H16, heptane O GH12, heptynearrow_forwardThe reduction of a certain alkene gave 2-methylbutane. It also gave acetic acid (CH3CO2H) as one of the products when treated with hot basic KMnO4 followed by acidification. Give the IUPAC name of this alkene.arrow_forward

 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



