
Concept explainers
Figure 4.6 Look at each of the processes shown and decide if it is endergonic or exergonic.

To analyze:
The process shown in the figure is endergonic or exergonic.
Introduction:
The endergonic process is the one which is accompanied by the absorption of energy from the surrounding, while exergonic processes is accompanied by the release of energy into the environment.
Explanation of Solution
Given:
Figure (a) 
Figure (b) 
Figure (c) 
Figure (d) 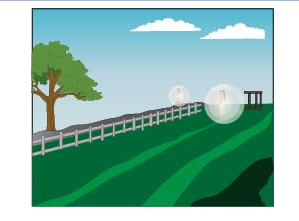
The first figure is of a compost. Decomposition of dead and decaying matter to produce compost or humus is an exergonic process because it is accompanied by the release of heat energy.
The second picture is of a chick coming out from an egg. It is an endergonic process because the process has taken in the energy from the chick.
The third picture is of tea dissolving in water, which is an exergonic process.
The last one is of a ball rolling downwards, in this case, the energy is dissipiated in the environment, which is an exothermic or exergonic process.
Decomposition, dissolving of tea in water and rolling of the ball are exergonic reactions, while the hatching of a chick from an egg is an example of an endergonic reaction.
Want to see more full solutions like this?
Chapter 4 Solutions
Concepts of Biology
Additional Science Textbook Solutions
College Physics
Biology: Life on Earth (11th Edition)
Fundamentals of Anatomy & Physiology Plus Mastering A&P with eText - Access Card Package (10th Edition) (New A&P Titles by Ric Martini and Judi Nath)
Campbell Essential Biology (7th Edition)
- Figure 6.8 Look at each of the processes shown, and decide if it is endergonic or exergonic. In each case, does enthalpy increase or decrease, and does entropy increase or decrease?arrow_forwardDescribe the ways an exergonic process can drive an endergonic process.arrow_forwardIdentify the two cellular process responsible for products described in the figure. Explain the role of each product listed in the processes. refer to picturearrow_forward
- Which process:arrow_forwardGive the products of each of the following processes. the words on the top will match up with the words on the bottom.arrow_forwardA reaction that has ΔH>0 and ΔS>0 is: exergonic if T < ΔH/ΔS. always exergonic. freely reversible. always endergonic. exergonic if T > ΔH/ΔS.arrow_forward
- Figure 4.5 Energy inputs and outputs in chemical reactions. 1 Some reactions convert molecules with lower energy to molecules with higher energy, so they require a net energy input in order to proceed. 2 Other reactions convert molecules with higher energy to molecules with lower energy, so they end with an energy release Figure It Out: Which law of thermodynamics explains energy inputs and outputs in chemical reactions?arrow_forwardIn an electric field,DNA moves from the ___to the ___ polearrow_forwardLabel the following statements true or false: (a) A reaction is said to be spontaneous when it can proceed in either the forward or reverse direction. (b) A spontaneous process always happens very quickly.arrow_forward
- Describe how vesicles are cycled in the presynaptic terminal. Use a diagram if necessaryarrow_forwardIn generating order within the cell and biological systems, explain on why does the cell does not defy the law of entropy.arrow_forwardThis reaction is exergonic because the reactants have more potential energy than the products.arrow_forward
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning




