
Concept explainers
Figure 4.15 Cyanide inhibits cytochrome c oxidase, a component of the electron transport chain. If cyanide poisoning occurs, would you expect the pH of the intermembrane space to increase or decrease? What affect would cyanide have on ATP synthesis?
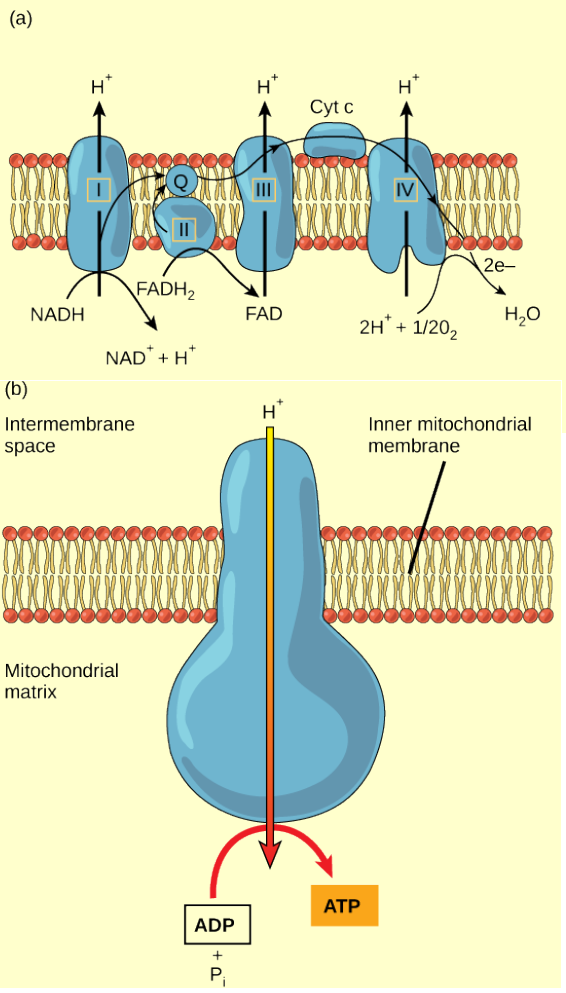
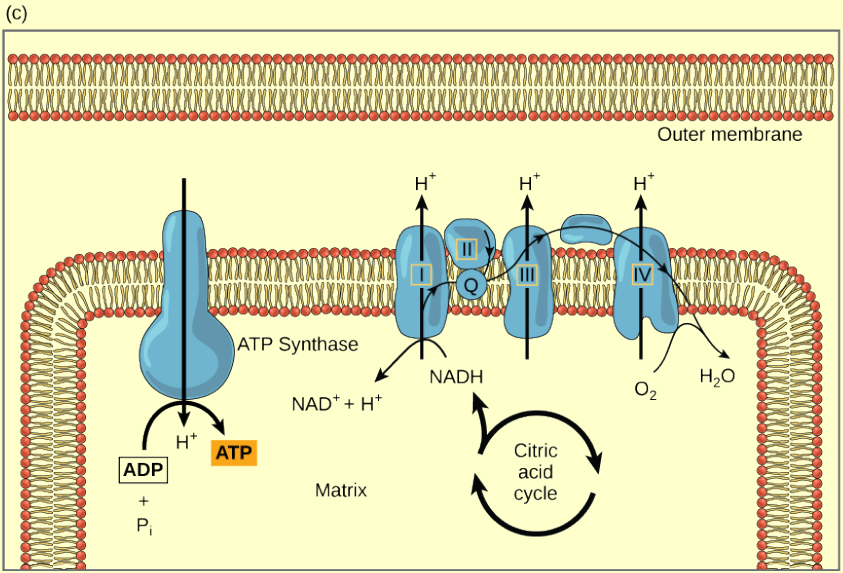
Figure 4.15 (a) The electron transport chain is a set of molecules that supports a series of
Trending nowThis is a popular solution!

Chapter 4 Solutions
Concepts of Biology
Additional Science Textbook Solutions
College Physics
Microbiology: An Introduction (13th Edition)
Genetic Analysis: An Integrated Approach (3rd Edition)
Genetic Analysis: An Integrated Approach (2nd Edition)
Campbell Essential Biology (7th Edition)
- Figure 7.12 Cyanide inhibits cytochrome c oxidase, a component of the electron transport chain. If cyanide poisoning occurs, would you expect the pH of the intermembrane space to increase or decrease? What effect would cyanide have on ATP synthesis?arrow_forwardWhich of the following statements about the generation of ATP in the electron transport chain is correct? a. The generation of ATP from ADP coupled to electron transfer occurs by substrate level phosphorylation as in glycolysis. b. ATP synthase generation of ATP involves a rotating structure inside the inner mitochondrial membrane. c. ATP synthase generation of ATP involves a rotating structure outside the inner mitochondrial membrane. d. Electron transport generates a proton gradient across the outer mitochondrial membrane.arrow_forwardWhich electron carrier would have the greatest negative impact on ATP production during oxidative phosphorylation if its production was inhibited? answer choices A.) Water B.) FADH2 C.) Oxygen D.) NADHarrow_forward
- Which of the following statements is TRUE of the electron transport chain (ETC)? A. The movement of electrons down the ETC drives the formation of a proton (H+) gradient. B. The movement of H+ ions through ATP synthase drives the transfer of electrons down the ETC. C. The movement of electrons down the ETC drives the movement of ADP and phosphate through ATP synthase. D. The movement of H+ ions across the inner membrane of the mitochondria drives the transfer of electrons down the ETC.arrow_forwardWhich of the following molecules remains oxidized when the electron transport inhibitor antimycin A is added to a suspension of actively respiring mitochondria? a. cytochrome c b. NAD c. cytochrome a d. UQarrow_forwardThe following statements describe the path of electrons from NADH through the electron transport chain. Please arrange them in order: Cytochrome c (Fe2+) carries electrons to Complex IV. Cytochrome c (Fe3+) is reduced to cytochrome c (Fe2+) QH2 carries electrons to Complex III Oxygen is reduced to water Ubiquinone (coenzyme Q) is reduced to ubiquinol (QH2)arrow_forward
- Figures A and B show ion H+ flow across membranes and ATP generation through membrane-bound ATP synthase. Which of the following best describes the specific gradients driving ATP synthesis in figures A and B? Figure A shows the proton gradient during oxidative phosphorylation in mitochondria, while figure B shows the proton gradient during photophosphorylation in chloroplasts. Figure A shows the NAD+ gradient during oxidative phosphorylation in mitochondria, while figure B shows the NADPH gradient during photophosphorylation in chloroplasts. Figure A shows the NAD+ gradient during the Krebs cycle and chemiosmosis in mitochondria, while figure B shows the NADPH gradient during the Calvin cycle and chemiosmosis in chloroplasts. Figure A shows the proton gradient during the Krebs cycle and chemiosmosis in mitochondria, while figure B shows the proton gradient during the Calvin cycle and chemiosmosis in chloroplasts.arrow_forwardWhich of the following drives the electron transfer from coenzyme Q to cytochrome c, which also results to the pumping of protons from the mitochondrial matrix to the intermembrane space? Complex I Complex II Complex III Complex IV If 2 molecules of FADH2 are oxidized during oxidative phosphorylation, how many molecules of ATP are synthesized? Your answer should be a numerical input.arrow_forwardWhich of the following statements about cellular respiration is TRUE in eukaryotes? A. For every molecule of NADH oxidized in the electron transport chain, 1 molecule of ATP is produced. B. ATP synthase harnesses the flow of protons (hydrogen ions = H+) from the mitochondrial matrix to the intermembrane space to produce ATP. C. ATP synthase transduces the flow of protons (hydrogen ions = H+) from the intermembrane space to the mitochondrial matrix into kinetic (mechanical) energy D. H2O is the final electron acceptor in the electron transport chain, being oxidized to O2 and H+. E. All of the above are truearrow_forward
- Where is ATP produced by oxidative phosphorylation? (where is it located immediately after the moment of synthesis) Group of answer choices the mitochondrial matrix the cytoplasm the intermembrane space the inner mitochondrial membrane Which of the following most readily donates electrons? (strongest reducing agent) Group of answer choices NADH NAD+ Ubiquinone lactate Protons first enter ATP synthase through which subunit? Group of answer choices β c γ aarrow_forwardWhich of the following statements is correct? a. Glycolysis and Krebs cycle produce ATP in substrate level phosphorylation b. Glycolysis and Krebs cycle produce ATP in oxidative level phosphorylation. c. Glycolysis and pyruvate oxidation produce ATP in substrate level phosphorylation. d. Glycolysis and pyruvate oxidation produce ATP in oxidative level phosphorylation.arrow_forwardWhich of the following is NOT true of the electron transport chain? a. It produces NADH, as a result of electrons being transferred from glucose to NAD+ b. End products include CO2, water, and pyruvate c. It allows for the controlled release of energy for ATP synthesis d. It is part of the oxidative phosphorylation processarrow_forward
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax


