
Concept explainers
Figure 4.5 Energy inputs and outputs in
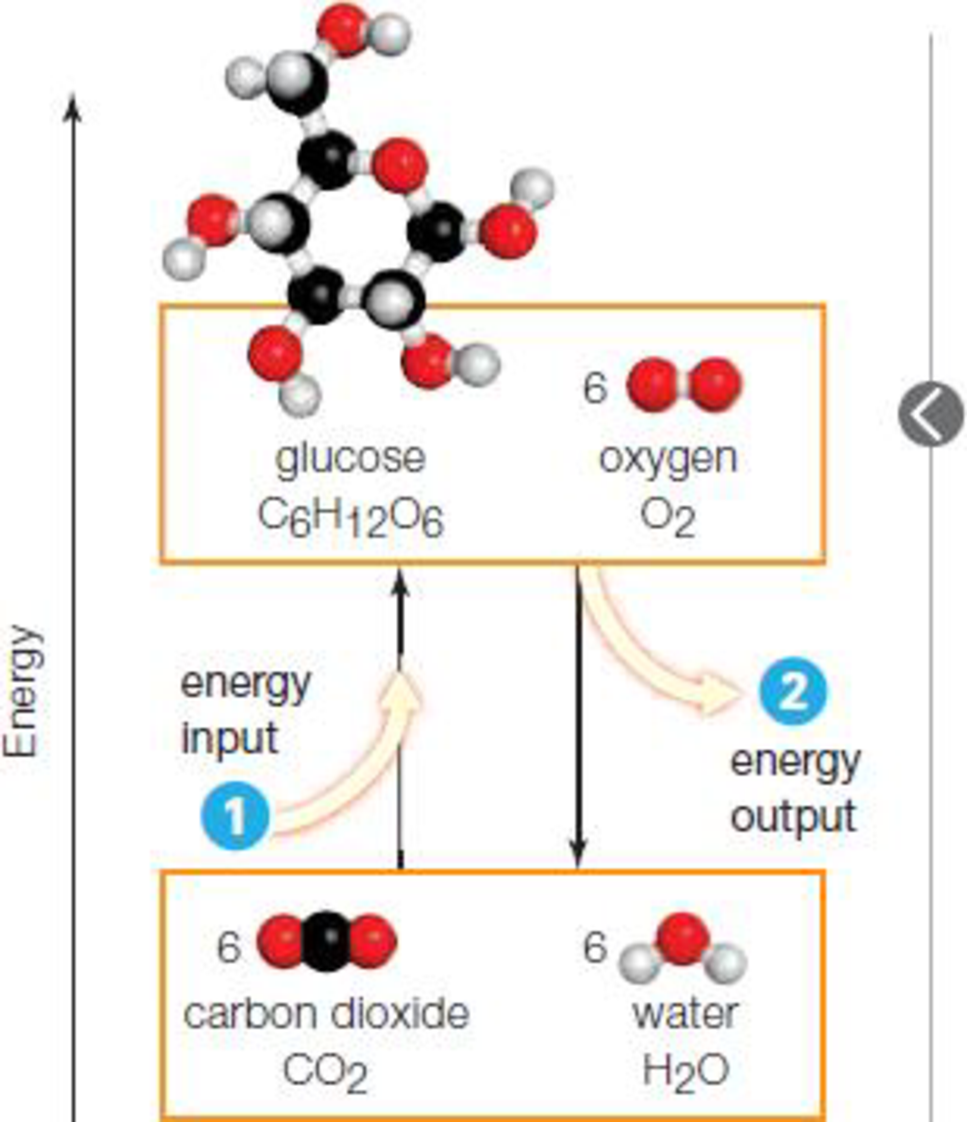
- 1 Some reactions convert molecules with lower energy to molecules with higher energy, so they require a net energy input in order to proceed.
- 2 Other reactions convert molecules with higher energy to molecules with lower energy, so they end with an energy release
Figure It Out: Which law of
To determine: The law of thermodynamics that explains the energy inputs and outputs in chemical reaction.
Introduction: Thermodynamics is defined as the study of heat as well as other forms of energy. In the term thermodynamics, therm indicates heat and dynam indicates power. The first law of thermodynamics states that energy can neither be created nor destroyed. The second law of thermodynamics states that the energy can spread out spontaneously.
Explanation of Solution
The first law of thermodynamics explains the energy inputs and energy outputs in the chemical reactions. The first law of thermodynamics is also called as the law of conservation of energy and it states that the energy can neither be created nor destroyed. The total amount of energy that is present before and after conversion remains the same. If the energy present in reactants is less than the product, then the reaction will proceed only with the net energy input. If the energy of the reactant is higher than the product, the reaction ends with a net energy release.
In the given figure, a lower energy molecule is converted to a higher energy molecule and for the reaction to proceed, it requires net energy input. In the other reaction, higher energy molecule is converted to the lower energy molecule with a net energy release. Thus, the first law of thermodynamics explains the input and output of energy in the given chemical reactions.
Want to see more full solutions like this?
Chapter 4 Solutions
Bundle: Biology Today And Tomorrow With Physiology, Loose-leaf Version, 5th + Lms Integrated For Mindtap Biology, 1 Term (6 Months) Printed Access Card
- The capacity to do work best defines: a metabolic pathway. entropy. kinetic or potential energy. a chemical equilibrium. thermodynamics.arrow_forwardWhich of the following statements is not correct? a. Energy cannot be created or destroyed. b. Energy cannot change from one form to another. c. Energy tends to disperse spontaneously. d. Energy can be transferred.arrow_forwardFigure 6.8 Look at each of the processes shown, and decide if it is endergonic or exergonic. In each case, does enthalpy increase or decrease, and does entropy increase or decrease?arrow_forward
- Which of the following statements is NOT true? None of the other four answers (all are true) The energy required to cause a chemical reaction to occur is called the activation energy Decomposition (degradation, catabolic) reactions are usually endergonic Chemical reactions in living cells are catalyzed by enzymes Enzymes increase the probability that a chemical reaction will take place by lowering the activation energyarrow_forwardYour body stores energy in the form of fat and other organic molecules. When your body needs extra energy, it breaks apart the molecules of fat to release that energy (sometimes called "burning fat"). That energy is used to power cellular reactions or is lost as heat. The total amount of energy used by your body or lost as heat equals the amount of energy the molecules of fat originally stored before breaking apart. This process is an example of the _________law of thermodynamics. A. first B. secondarrow_forwardItem 2 The propagation of an impulse along the axon of a neuron is considered electrical energy, a form of kinetic energy. True or False Item3 Item 3 Stored energy is described as potential energy. True or False Item4 Item 4 __________ is a monomer that is stored as the polymer glycogen within the liver and muscle. Fill in the blank Item5 Item 5 The first law of thermodynamics states that once energy is consumed, it can no longer be converted to any other form of energy. True or False Item6 Item 6 As a runner sprints down a track, her movement is an example of Multiple Choice potential energy. mechanical energy. heat. radiant energy. chemical energy. Item7 Item 7 Which is a form of energy that is generally unavailable to do any work? Multiple Choice Heat Kinetic energy Radiant energy Sound energy Mechanical energy Item8 Item 8 Three molecules that are…arrow_forward
- Use the following graph to diagram the energetics of a chemical reaction, with and without an enzyme. Be sure to position reactants and products at appropriate points and to indicate the stages in the reaction and the energy levels.arrow_forwardThe movement of energy between living organisms is a matter of atoms, molecules and compounds being built into larger molecules or being broke into smaller ones. Reviewing the two equations for energy transference, which one is building energy? 6CO2 + 6H2O + energy --------> C6H12O6 + 6O2 C6H12O6 + 6O2 ---------> 6CO2 + 6H2O + energyarrow_forwardHow it is that cells are making larger, more complex molecules, yet they do not defy the second law of thermodynamics? (note: cells are open systems not closed)arrow_forward
- The graph below shows how temperature affects the rate of chemical reactions At what temperature is the rate of reaction the highest?arrow_forwardThe oxidation of sugar molecules by the cell takes place according to the general reaction C6H12O6 (glucose) + 6O2 → 6CO2 + 6H2O + energy. which of the following statements are correct? explain your answers. all of the energy produced is in the form of heat. none of the produced energy is in the form of heat. The energy is produced by a process that involves the oxidation of carbon atoms. The reaction supplies the cell with essential water. in cells, the reaction takes place in more than one step. many steps in the oxidation of sugar molecules involve reaction with oxygen gas. some organisms carry out the reverse reaction. some cells that grow in the absence of O2 produce CO2.arrow_forwardenzymes increase the rate of a chemical reaction by increasing the amount of activation energy needed decreasing the amount of activation energy needed decreasing the temperature increasing the temperature altering the chemical of substrates 2. which of the following is an example of quantitive data? the freshman class of the students contained 643 students, 372 males, and 271 females. the absorption peak of cobalt chloride was 510nm bromelain has optimal enzyme activity at a pH of 7 the color of cobalt chloride was pink the mass of the onion was 50 gramsarrow_forward
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
 Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning




