
EBK CAMPBELL BIOLOGY
10th Edition
ISBN: 9780136539414
Author: Reece
Publisher: VST
expand_more
expand_more
format_list_bulleted
Question
Chapter 4, Problem 2TYU
Summary Introduction
To find: The functional group not present in the Fig.1.
Pictorial representation: The molecule in the Fig.1 represents different
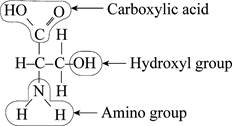
Fig.1: A molecule
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Functional group as indicated by letters A,B,C and D?
OFF
Draw with
Add Pen
Trackpad
Q1: Complete the following table:
(A.C 1.1, 1.2)
Molecular
Formula
Molecule
Structural Formula
o-d=0
CH2
CH3
Variable
H.
--H-N
H.
H.
H
1--
Amino Acid
C2H5NO2
(Glycine R=H)
Glycerol
H H
.
H H
HH
H H
но
.
H H
H H
H HH
a-glucose
glish (United Kingdom)
MacBook Pro
Macmillan Learning
Consider the structure of methionine in its +1 charge state.
H₂N
CH₂
CH₂
CH3
-OH
Give the pK, value for the a-amino group of methionine. An
answer within ±0.5 units is acceptable.
Give the pK, value for the a-carboxyl group of methionine.
An answer within +0.5 units is acceptable.
Determine the isoelectric point (pl) for methionine. Give your
answer to two decimal places.
pK (−NH ) =
pK₂ (-COOH) =
pl =
5.74
2.28
Incorrect
9.21
Incorrect
Attempt
Chapter 4 Solutions
EBK CAMPBELL BIOLOGY
Ch. 4.1 - Why was Whler astonished to find he had made urea?Ch. 4.1 - VISUAL SKILLS See Figure 4.2. Miller carried out...Ch. 4.2 - DRAW IT (a) Draw a structural formula for C2H4....Ch. 4.2 - Prob. 2CCCh. 4.2 - How are gasoline and fat chemically similar?Ch. 4.2 - VISUAL SKILLS See Figures 4.5a and 4.7. Can...Ch. 4.3 - VISUAL SKILLS What does the term amino acid...Ch. 4.3 - What chemical change occurs to ATP when it reacts...Ch. 4.3 - DRAW IT Suggose you had an organic molecule such...Ch. 4 - How did Stanley Miller's experiments support the...
Ch. 4 - Prob. 4.2CRCh. 4 - In what ways does a methyl group differ chemically...Ch. 4 - Organic chemistry is currently defined as (A) the...Ch. 4 - Prob. 2TYUCh. 4 - MAKE CONNECTIONS Which chemical group is most...Ch. 4 - VISUAL SKILLS Visualize the structural formula of...Ch. 4 - visual skills Choose the term that correctly...Ch. 4 - VISUAL SKILLS Identify the asymmetric carbon in...Ch. 4 - Which action could produce a carbonyl group? (A)...Ch. 4 - Prob. 8TYUCh. 4 - Prob. 9TYUCh. 4 - SCIENTIFIC INQUIRY 50 years ago, pregnant women...Ch. 4 - WRITE ABOUT A THEME: ORGANIZATION In 1918, an...Ch. 4 - SYNTHESIZE YOUR KNOWLEDGE Explain how the chemical...
Knowledge Booster
Similar questions
- For the tripeptide: methionylisoleucylcysteine (i) draw the structure of each tripeptide (ii) label the amide bonds in your structures (iii) identify the N-terminal and C-terminal amino acidsarrow_forwardFunctional Group Name Structural Diagram (draw all bonds) Found where in the body??? Hydroxyl -N H Carbonyl #1 (aldehydes)arrow_forwardIdentify. Examine the following four amino acids (A-D): Co0 "H,N- CH "H,N CH "H;N-CH "H,N CH CH2 CH2 CH, CH CH2 CH3 CH, CH2 OH NH," B D What are their names, three-letter abbreviations, and one-letter symbols?arrow_forward
- B-pt and M-pt of various organic compounds depends on intermolecular forces of attraction which depend on following (a) Inter molecular/intramolecular H-bond. (b) Dipole-Dipole interaction (Carboxyl and ether). (c) Molecular size (d) Surface area. (branching).arrow_forwardA monosaccharide designated as an aldehyde sugar contains(a) a terminal carboxyl group (b) an internal carboxyl group(c) a terminal carbonyl group (d) an internal carbonyl group(e) a terminal carboxyl group and an internal carbonyl grouparrow_forwardBIOCHEMISTRY LECTURE SOLUTIONS AND ANSWERS SHOULD BE HANDWRITTEN. : The following structure is sorbose CH2OH C = O Н-С-ОН НО - С - Н 4. Draw the boat conformation of no. 3. Н-С-ОН 5. Draw the chair conformation of no. 3. CH2OH 6. How many stereoisomers does sorbose have? 7. Draw all the L isomers of this sugar. 8. Group the L isomers as enantiomers and diastereomers. 9. Draw B-D-sorbofuranose. Draw B-D-sorbofuranosyl(2–1) B-D-glucopyranoside. Is this a reducing sugar? What would be the probable function of this disaccharide, structural or storage?arrow_forward
- Which of the following are saponifiable lipids? (Recall that ester bonds are broken by base hydrolysis.)(a) Progesterone (b) Glyceryl trioleate(c) A sphingomyelin (d) Prostaglandin E1(e) A cerebroside (f) A lecithinarrow_forwardCalculate the charge and draw the structure of the predominant ion for: (i) Lysine at pH 7.0 (R=-CH2CH;CH;CH¿NH2) (ii) Glutamic acid at pH 7.0 (R=-CH2CH;COOH) (iii) Serine at pH 1.0 (R=-CH2OH)arrow_forwardCompare the stability of the two salt bridges shown circled in blue at pH 8.4. The carboxylate ions are completely de- protonated at pH 8.4 The pka of the amino group (upper left) is 8.1 and the pka of the amino group (bottom right) is 8.7. (d) NH, F-8- Ed (b) O-H- M5 CH₂COOH (c) 5 (b) CH, CH. H--O=C (d) -H H CH,OH (b) KCHJ NH, (a) The bottom right salt bridge is stronger due to the partial positive charge of +2/3. The bottom right salt bridge is stronger due to the partial positive charge of +1/3. The upper left salt bridge is stronger due to the partial positive charge of +1/3. The upper left salt bridge is stronger due to the partial positive charge of +2/3.arrow_forward
- dentify (showing clearly the basis for your answer in each case) which one(s) can serve as, (a) H-bond donor b) H-bond acceptorarrow_forward(c) The i-motif structure requires a pH of between 4.2 and 5.2. (i) Explain, using chemical structures, why this is the case.arrow_forwardLearning Task 2. Letl's Categorize! On a separate sheet of paper, given the structural formula, complete the table by providing the needed information regarding the different biomolecules. Biomolecule Building blocks Classification or Types Structural Formula / model Smgle ta CH,OH Amino Acid Structure Hydrogen Amino Carboxyl H R R-group (variant)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning