
Concept explainers
The sketch of the graph for the given function and to explain the trends in the graph.
Answer to Problem 4.1P
The graph for the function
Explanation of Solution
Given:
The Planck distribution function for blackbody
Calculation:
Consider the term,
The value of the term is calculated as,
Consider the term,
The value of the term is calculated as,
Thus, the Planck's distribution function of a blackbody radiation for a fixed temperature
The following table shows the Planck's intensity distribution for different wavelengths at a fixed temperature.
| Wavelength | |
The following figure shows the Planck's intensity distribution for different wavelengths at a fixed temperature.
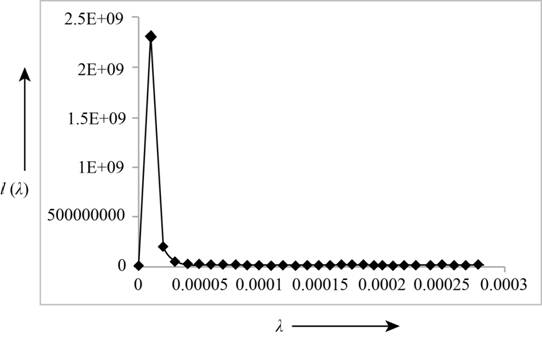
Figure (1)
From the graph, it can be concluded that the intensity of blackbody radiation for a fixed temperature, is maximum at a particular wavelength and it becomes zero with the increase in wavelength. With the increase in temperature, the maximum intensity of a radiation shift towards the shorter wavelengths.
Conclusion:
Therefore, the graph for the function
Want to see more full solutions like this?
Chapter 4 Solutions
Modern Physics For Scientists And Engineers
- What are (a) the energy of a photon corresponding to wavelength 1.00 nm, (b) the kinetic energy of an electron with de Broglie wavelength 1.00 nm, (c) the energy of a photon corresponding to wavelength 1.00 fm, and (d) the kinetic energy of an electron with de Broglie wavelength 1.00 fm?arrow_forwardWhat is the wavelength of (a) a photon with energy 1.00 eV, (b) an electron with energy 1.00 eV, (c) a photon of energy 1.00 GeV, and (d) an electron with energy 1.00 GeV?arrow_forwardA blackbody (a hollow sphere whose inside is black) emits radiation when it is heated. The emittance (Mλ, W/m3), which is the power per unit area per wavelength, at a given temperature (T, K) and wavelength (λ, m) is given by the Planck distribution, where h is Planck's constant, c is the speed of light, and k is Boltzmann's constant. Determine the temperature in degrees Celsius at which a blackbody will emit light of wavelength 3.57 μm with an Mλ of 5.31×1010 W/m3. The power per unit area emitted can be determined by integrating Mλ between two wavelengths, λ1 and λ2. However, for narrow wavelength ranges (Δλ), the power emitted can be simply calculated as the product of Mλ and Δλ. power emitted=MλΔλ Using the conditions from the first part of the question, determine the power emitted per square meter (W/m2) between the wavelengths 3.56 μm and 3.58 μm.arrow_forward
- As noted in the chapter, the cosmic microwave background radiation fits the Planck equation for a blackbody at 2.7 K. (a) What is the wavelength at the maximum intensity of the spectrum of the background radiation? (b) What is the frequency of the radiation at the maximum? (c) What is the total power incident on Earth from the background radiationarrow_forwardWhich of the following statements is true regarding how blackbody radiation changes as the temperature of the radiating object increases? 1. Both the maximum intensity and the peak wavelength decrease. 2. Both the maximum intensity and the peak wavelength increase. 3. The maximum intensity increases, and the peak wavelength decreases. 4. The maximum intensity decreases, and the peak wavelength increases.arrow_forwardWhich statement is TRUE about Einstein’s explanation regarding the photoelectric effect? I. The effect is not observed below a certain cut-off frequency since the photon energy must be greater than or equal to the work function.II. The maximum KE depends only on the frequency and the work function.III. The maximum KE depends only on the frequency, work function and intensity of light.IV. The maximum KE increases with increasing frequency. a) I, II and IV b) I and III c) I, II and III d) I and IIarrow_forward
- Which of the following statements about a black body are true? Select one or more: a.The spectrum of the cosmic background radiation corresponds with great accuracy to the radiation of a black body at a temperature of 2.7 K. b.A black body absorbs all the radiation that hits it, and emits no radiation at all. c.According to Planck's radiation law (black body distribution), the wavelength corresponding to the maximum energy density of the radiation decreases (and the frequency increases) as the temperature increases. d.A black body reflects all the radiation that hits it, and absorbs no radiation at all.arrow_forwardIf a body is at 2000 K, the wavelength at which the body emits the maximum amount of radiation is a.) 1.45 um b.) 1.45 cm c.) 0.345 cm d.) 0.345 umarrow_forwardThe mass of a proton is 1.67 × 10-27 kg. If a proton has the same momentum as a photon with a wavelength of 325 nm, what is its speed? a. 2.73 × 10-3 m/s b. 0.819 m/s c. 1.22 m/s d. 2.71 × 104 m/sarrow_forward
- Visible light falls into wavelength ranges of 400-700 nm, for which 1 m=1×109 nm The energy and wavelength of light are related by the equationE=hcλ where E is energy in Joules, h is Planck's constant ( 6.626×10−34 J-s ), c is the speed of light ( 2.998×108 m/s), and λ is the wavelength in m. If a visible light photon has a wavelength of 632.3 nm, what is the energy of the photon (in J)?arrow_forwardA certain shade of blue has a frequency of 7.32×1014 Hz.7.32×1014 Hz. What is the energy of exactly one photon of this light? Planck's constant is ℎ=6.626×10−34 J⋅s. E=?arrow_forwardAn electron has a non-relativistic speed of v=1.5 x 105 m/s. Determine: a) Its De Broglie wave length, its kinetic energy and the rest energy of the electron in joules and electron volts b) What is the speed of the electron if the relativistic kinetic energy is equal to the rest energy? c) The momentum of the electron at a speed of 0.80c and the De Broglie wavelength for an electron with a speed of 0.80c.arrow_forward
 College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning
Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning




