
Concept explainers
To determine:
The difference between a Lewis structure and a condensed structure in terms of atoms and bonds is shown in the structure.
Introduction:
The molecules of organic compounds are represented by four different ways namely, Lewis structure, skeletal formula, condensed structural formula, and ball-and-stick model.
Lewis structures show all atoms, bonds, and lone pairs of atoms in the molecule. It helps in predicting the molecular geometry and shows the connectivity of atoms in a molecule whereas, condensed structural formula is simpler as it does not show any bonds or lone pairs of atoms and shows no connectivity of atoms in a molecule.
Explanation of Solution
The example of butanol molecule is to be assumed.
The Lewis structure of butanol molecule is represented as,
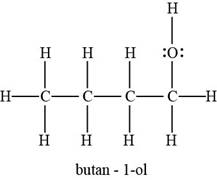
Fig.1
The number of bonds of each atom depends on the valence electrons of atoms in a molecule.
Figure 1 shows the Lewis structure in which carbon has
Condensed structure shows the shorter way to draw a molecule and is more simplified than the Lewis structure. It shows all atoms except the vertical bonds and most or all the horizontal bonds.
The condensed structure of butanol molecule is represented as,
In this formula, first carbon atom has three hydrogen atoms bonded to it; the second, third, and fourth carbon has two hydrogen atoms. The two lone pair on oxygen atoms are not shown as in condensed structure of atoms. Lone pair is not represented in the chemical symbol of atom.
Lewis structure is the most expanded structure as it shows all atoms, bonds, and lone pairs of atoms in a molecule whereas, condensed structure is the least expanded as it does not show all the bonds and lone pairs of atoms. Condensed structure is simpler than the Lewis structure.
Want to see more full solutions like this?
Chapter 4 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Write the condensed structures of all the isomers of octane.arrow_forward||| O NAMING AND DRAWING ORGANIC MOLECULES Interpreting condensed chemical structures Use this condensed chemical structure to complete the table below. 0 NH₂ - CH — C — OH | CH3 The condensed chemical structure of alanine Some facts about the alanine molecule: number of carbon-carbon single (C - C) bonds: number of carbon-hydrogen single (C-H) bonds: number of oxygen-hydrogen single (O - H) bonds: number of nitrogen-hydrogen single (N - H) bonds: number of lone pairs: X Ś 0 ☐ 1 D 503 Jessarrow_forwardDefine the condensed structure of Compounds Containing a C=O Group ?arrow_forward
- draw the condensed structure of 2,3-dimethyl -1 pentanol ?arrow_forwardDraw the condensed structure of an isomer of this molecule: CH3 C—C—O-CH3arrow_forwardFrom the condensed structural formulas,draw out the expanded structural formula for each in the table below(include all hydrogen). Name the organic compound using International union of pure and applied chemistry rules.arrow_forward
- Structure and Bonding Interpreting condensed chemical structures Use this condensed chemical structure to complete the table below. O CH2=CH-C-H The condensed chemical structure of acrolein Some facts about the acrolein molecule: number of carbon-carbon single (CC) bonds: number of carbon-hydrogen single (CH) bonds: number of lone pairs:arrow_forwardWhat is the condensed structure of Tertbutyl cyclohexanearrow_forwardChoose the functional group represented by the condensed structural formula (CH3)2CHCHO.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





