
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.30P
Will acetylene react with sodium hydride according to the following equation to form a salt and hydrogen, H2? Using pKa values given in Table 4.1, calculate Keq for this equilibrium.
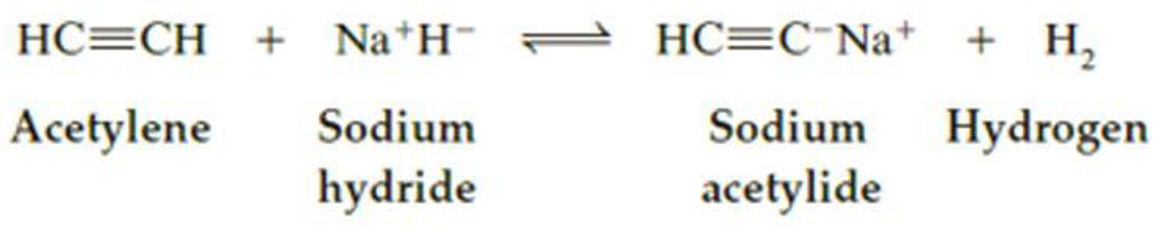
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Provide an explanation without using the pka values :
Why is phenol stronger acid than butanoic acid?
How does the first step in the reaction of propene with Br2 differ from the first step in the reaction of propene with HBr?
Give one example for the hydration of Alkenes: that is: the addition of H2O by Hydroboration.
Chapter 4 Solutions
Organic Chemistry
Ch. 4.2 - For each conjugate acid-base pair, identify the...Ch. 4.2 - Write these reactions as proton-transfer...Ch. 4.2 - Following is a structural formula for guanidine,...Ch. 4.2 - Write an equation to show the proton transfer...Ch. 4.3 - For each value of Ka, calculate the corresponding...Ch. 4.4 - Predict the position of equilibrium and calculate...Ch. 4.5 - Calculate Keq for a reaction with G0 = 17.1 kJ/mol...Ch. 4.6 - Acid-Base Equilibria Many factors contribute to...Ch. 4.6 - What is the relative trend in acidity and pKa of...Ch. 4.7 - Write an equation for the reaction between each...
Ch. 4 - For each conjugate acid-base pair, identify the...Ch. 4 - Complete a net ionic equation for each...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Prob. 4.12PCh. 4 - In acetic acid, CH3COOH, the OH hydrogen is more...Ch. 4 - Which has the larger numerical value? (a) The pKa...Ch. 4 - In each pair, select the stronger acid. (a)...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - If the G for a reaction is 4.5 kcal/mol at 298 K,...Ch. 4 - Calculate the Keq for the following reactions from...Ch. 4 - Prob. 4.20PCh. 4 - Answer true or false to the following statements...Ch. 4 - In each of the following three reaction coordinate...Ch. 4 - The acid-base chemistry reaction of barium...Ch. 4 - Unless under pressure, carbonic acid (H2CO3) in...Ch. 4 - Prob. 4.25PCh. 4 - Acetic acid, CH3COOH, is a weak organic acid, pKa...Ch. 4 - Benzoic acid, C6H5COOH (pKa 4.19), is only...Ch. 4 - Prob. 4.28PCh. 4 - One way to determine the predominant species at...Ch. 4 - Will acetylene react with sodium hydride according...Ch. 4 - Prob. 4.31PCh. 4 - For each equation, label the Lewis acid and the...Ch. 4 - Complete the equation for the reaction between...Ch. 4 - Each of these reactions can be written as a Lewis...Ch. 4 - The sec-butyl cation can react as both a...Ch. 4 - Prob. 4.36APCh. 4 - Prob. 4.37APCh. 4 - Prob. 4.38APCh. 4 - Explain why the hydronium ion, H3O+, is the...Ch. 4 - What is the strongest base that can exist in...Ch. 4 - Prob. 4.42APCh. 4 - Prob. 4.43APCh. 4 - Methyl isocyanate, CH3N=C=O, is used in the...Ch. 4 - Offer an explanation for the following...Ch. 4 - Prob. 4.46APCh. 4 - Alcohols (Chapter 10) are weak organic acids, pKa...Ch. 4 - As we shall see in Chapter 19, hydrogens on a...Ch. 4 - 2,4-Pentanedione is a considerably stronger acid...Ch. 4 - Write an equation for the acid-base reaction...Ch. 4 - Prob. 4.51APCh. 4 - Prob. 4.52APCh. 4 - Prob. 4.53APCh. 4 - Following is a structural formula for imidazole, a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write an equation for the reaction of chloroacetic acid (Ka=1.5103) with trimethylamine (Kb=5.9105) . Calculate the equilibrium constant for the reaction. If 0.10 M solutions of these two species are mixed, what will be their concentrations at equilibrium?arrow_forwardWhen aniline, C6H5NH2(Kb=7.41010) , reacts with a strong acid, its conjugate acid, C6H5NH3+, is formed. Calculate the pH of a 0.100 M solution of C6H5NH3+ and compare it with the pH of acetic acid (Ka=1.86105) .arrow_forwardExplain the reaction between Methyl Salicylate with acetyl chloride (CH3COCl). Gives your answer in detail from the name of the reaction until the formation of the product. You are advised to give an illustration of the reaction.arrow_forward
- Give the structural equation for the formation of the ester methyl butanoate a condensation reaction. Include necessary conditions. Identify the small molecule byproduct and show where the atoms in that byproduct came from. Indicate the new bond in the ester product. Give possible reactants to form the following condensation products and side products heat H + +H-CI Harrow_forwardDetermine the percent yield for an acylation reaction if beginning with 1.12 g of salicylic acid as the limiting reagent and 1.25 g of product was recovered. If 1.3 g salicylic acid and 0.8 mL acetic anhydride are used in the synthesis of aspirin, determine which is the limiting reagent and the theoretical yield..arrow_forward1. Why is the internal use of methyl salicylate limited to small quantities only? 2. What are the uses and application of methyl salicylate? 3. What is the role of salicylic acid, sulfuric acid and sodium carbonate in the synthesis of methyl salicylate? 4. What are the common methods to extract methyl salicylate from natural sources?arrow_forward
- Trimyristin is a fatty acid ester of my rustic acid and glycerol. Draw the structure of the products of the base-catalyzed hydrolysis of trimyristin. Using this reaction, calculate a theoretical yield if you started with 1 g of trimyristin.arrow_forwardCreate balanced chemical reactions for the following reactions. Organic reactions should be written with the line-angle drawing of each structure. 1. Reacting salicylic acid with acetic anhydride to form acetylsalicylic acid, with the use of sulfuric acid as a catalyst. 2. Water reacting with acetic anhydride to form acetic acid.arrow_forward3. Consider the pictured acid-catalyzed esterification. a) What is the limiting reagent in the reaction? and explain how you chose the limiting reagent, including any performed calculations. b) What is the theoretical yield (in g) of octyl acetate? c) Assume that your isolated yield of octyl acetate was 5.00 mL, calculate the percent yield of the reaction.arrow_forward
- Write the equation for the chemical reaction below. Addition of HBr with peroxide to pentenearrow_forwardCalculate the percent yield and theoretical yield for the following reaction: oxidation of cyclododecanol to cyclododecanone. 0.2093 grams of Cyclododecanol 0.5 mL of acetone 0.20 mL of Glacial acetic acid 2.0 mL of bleach Final weight of cyclododecanone = 0.0578 garrow_forwardPotassium permanganate and potassium dichromate are very similar in their oxidizing abilities, however there are differences. If I want to convert 4-hexen-1-ol into 4-hexenoic acid, which would be the appropriate oxidizing agent to use? Explain your answer using equations that show the two different products that would form via the two different oxidizing agents.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY