
Concept explainers
(a)
Interpretation: The explanation corresponding to the incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The naming of substituents attached to the longest carbon chain is not in alphabetical order. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
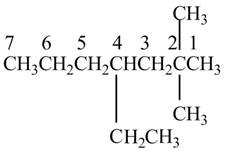
Figure 1
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
The given name is
The naming of substituents attached to the longest carbon chain is not in alphabetical order. The correct IUPAC name is
(b)
Interpretation: The explanation corresponding to the incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The longest carbon chain in the given IUPAC name contains seven carbon atoms instead of six carbon atoms. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
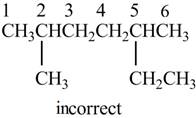
Figure 2
According to the given IUPAC name, the structure contains six carbon atoms.
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Thus, the correct structure for the given name is shown below.
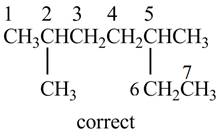
Figure 3
The longest carbon chain in the given IUPAC name contains seven carbon atoms instead of six carbon atoms. Thus, the correct IUPAC name is
The longest carbon chain in the given IUPAC name contains seven carbon atoms instead of six carbon atoms., the correct IUPAC name is
(c)
Interpretation: The explanation corresponding to the given incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The longest carbon chain in the given IUPAC name contains eight carbon atoms instead of seven carbon atoms. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
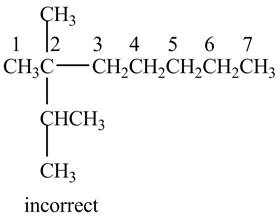
Figure 4
According to the given IUPAC name, the structure contains seven carbon atoms.
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Thus, the correct structure for the given name is shown below.
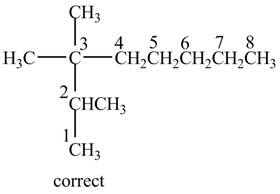
Figure 5
The longest carbon chain in the given IUPAC name contains eight carbon atoms instead of seven carbon atoms. Therefore, the correct IUPAC name is
The longest carbon chain in the given IUPAC name contains eight carbon atoms instead of seven carbon atoms. The correct IUPAC name is
(d)
Interpretation: The explanation corresponding to the given incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The structure corresponding to the given name is incorrect because the numbering of substituents is wrong. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
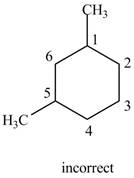
Figure 6
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
The given name is
The structure corresponding to the given name is incorrect because higher number is assigned to the second methyl substituent.
Therefore, the correct structure is shown below.
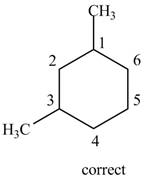
Figure 7
Thus, the correct IUPAC name is
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent. The correct IUPAC name is
(e)
Interpretation: The explanation corresponding to the incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
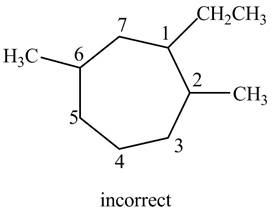
Figure 8
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
The given name is
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent.
Therefore, the correct structure is shown below.
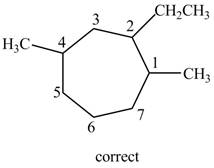
Figure 9
Thus, the correct IUPAC name is
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent. The correct IUPAC name is
(f)
Interpretation: The explanation corresponding to the incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The structure corresponding to the given name is incorrect because the numbering of substituents is wrong. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
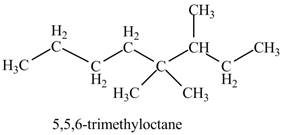
Figure 10
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
The given name is
The structure corresponding to the given name is incorrect because substituents should get lowest locant number.
Therefore, the correct structure is shown below.
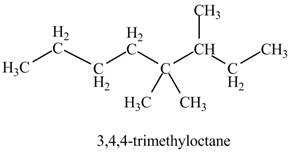
Figure 11
Thus, the correct IUPAC name is
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent. The correct IUPAC name is
(g)
Interpretation: The explanation corresponding to the incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The structure corresponding to the given name is incorrect because the numbering of substituents is wrong. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
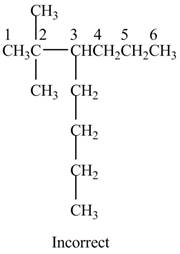
Figure 12
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
The given name is
Therefore, the correct structure is shown below.
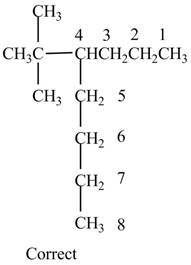
Figure 13
Thus, the correct IUPAC name is
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent. The correct IUPAC name is
(h)
Interpretation: The explanation corresponding to the incorrect IUPAC name
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
4. First identify the word root for the given compound.
5. The suffix used in the compound like –ene.
6. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 4.43P
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent. The correct IUPAC name is
Explanation of Solution
The given IUPAC name is
The structure corresponding to the given IUPAC name is shown below.
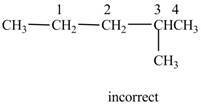
Figure 14
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
The given name is
Therefore, the correct structure is shown below.
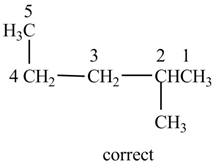
Figure 15
The longest carbon chain contains five carbon atoms instead of four carbon atoms.
Thus, the correct IUPAC name is
The structure corresponding to the given name is incorrect because higher number is assigned to the second substituent. The correct IUPAC name is
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry - With Access (Custom)
- Determine which part/s of the following IUPAC name made them incorrect; locant, parent length, prefix, none 3 - methylbutane 5-ethyl-4-propyloctane 1-(1-methylpentyl)cyclopentane 4,4 - di(methylethyl)heptane 1,1-methylbicyclo(1.1.0)butane 3-propylspiro(2.2)pentane 4-ethyl-5-propyloctanearrow_forward(a) 1-chloropropane (b) 2-chloropropane(c) 2,2-dimethylbutane (d) 2,3-dimethylbutane(e) 1-bromo-4-methylbenzene (f) 1-bromo-2-methylbenzene,arrow_forwardExplain why each name is incorrect, and then write a correct name. (a) 2-Ethyl-1-propene (b) 4-Methyl-4-hexanearrow_forward
- Draw the structure corresponding to each IUPAC name. a. 2,3,4,5−tetramethyldecane draw structure ... b. cyclononane draw structure ...arrow_forward1. What is the correct IUPAC name for 5-butyl-6-ethyl-4-methylhex-1-ene (For Alkene and Alkyne Naming, follow this format “Branch-Carbon Prefix-Functional Group Suffix”) e.g. 3-methylhex-2-ene 2. What is the correct IUPAC name for 3,4-dimethylhex-5-ene-1-ynearrow_forwardWhat is the correct IUPAC or systematic name for this compound? a. 3-methylbutane b. 3-methylpentane c. 2-methylbutane d. pentane e. 2-methylpentanearrow_forward
- Give the structure corresponding to each IUPAC name. a. 3-methylhexane c. 3,5,5-trimethyloctane b. 3,3-dimethylpentane d. 3-ethyl-4-methylhexanearrow_forwardDraw the structure corresponding to each IUPAC name. a.3-ethyl-2-methylhexane b. sec-butylcyclopentane c.4-isopropyl-2,4,5-trimethylundecane d.cyclobutylcycloheptane e.3-ethyl-1,1-dimethylcyclohexane f. 4-butyl-1,1-diethylcyclooctane g.6-isopropyl-2,3-dimethyldodecane h. 2,2,6,6,7-pentamethyloctane i. cis-1-ethyl-3-methylcyclopentane j. trans-1-tert-butyl-4-ethylcyclohexanearrow_forwardGive the structure corresponding to each IUPAC name. a. 3-methylhexane b. 3,3-dimethylpentane c. 3,5,5-trimethyloctane d. 3-ethyl-4-methylhexane e. 3-ethyl-5-isobutylnonanearrow_forward
- The IUPAC name 4-hexylhexane is incorrect. What should be the correct name? Group of answer choices 3-hexylhexane 7-ethyldecane 3-propylnonane 4-ethyldecanearrow_forwardWhat is the IUPAC name of this molecule? a. 1-chloropentane b. 2-chloro-1-ethylethane c. 1-chloro-2-ethylethane d. 1-chlorobutane e. non of these are correct.arrow_forwardExplain why each name is incorrect and then write a correct name. (c) 2-Methylcyclohexene (d) 3,3-Dimethylpentene (f) 2-Isopropyl-2-butenearrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





