
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
8th Edition
ISBN: 9781305864504
Author: Brent L. Iverson, Sheila Iverson
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.50AP
Write an equation for the acid-base reaction between 2,4-pentanedione and sodium ethoxide and calculate its equilibrium constant, Keq. The pKa of 2,4-pentanedione is 9; that of ethanol is 15.9.
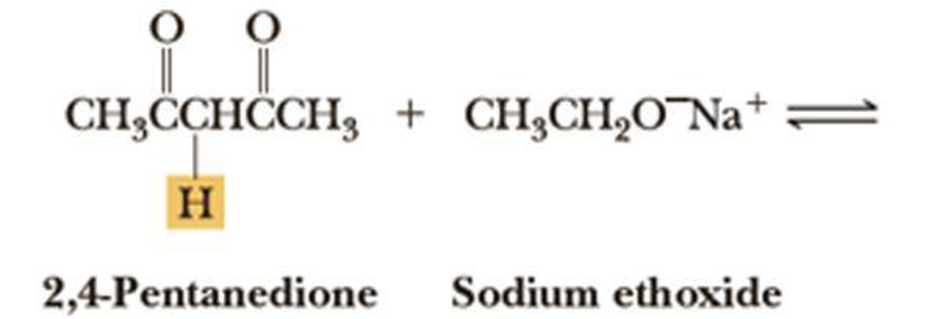
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Aldehydes and ketones undergo condensation reaction with 2, 4-dinitrophenylhydrazine. Write an equation for the reaction of 2, 4-dinitrophenylhydrazine with any one of the pheromones.
What makes 2, 4-dinitrophenylhydrazine suitable for characterizing aldehydes and ketones?
Account the solubility of carboxylic acids in distilled water
Identify the principal organic product in each case
NaH
CH3C
.OH
7
Chapter 4 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Ch. 4.2 - For each conjugate acid-base pair, identify the...Ch. 4.2 - Write these reactions as proton-transfer...Ch. 4.2 - Following is a structural formula for guanidine,...Ch. 4.2 - Write an equation to show the proton transfer...Ch. 4.3 - For each value of Ka, calculate the corresponding...Ch. 4.4 - Predict the position of equilibrium and calculate...Ch. 4.5 - Calculate Keq for a reaction with G0 = 17.1 kJ/mol...Ch. 4.6 - Acid-Base Equilibria Many factors contribute to...Ch. 4.6 - What is the relative trend in acidity and pKa of...Ch. 4.7 - Write an equation for the reaction between each...
Ch. 4 - For each conjugate acid-base pair, identify the...Ch. 4 - Complete a net ionic equation for each...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Prob. 4.12PCh. 4 - In acetic acid, CH3COOH, the OH hydrogen is more...Ch. 4 - Which has the larger numerical value? (a) The pKa...Ch. 4 - In each pair, select the stronger acid. (a)...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - If the G for a reaction is 4.5 kcal/mol at 298 K,...Ch. 4 - Calculate the Keq for the following reactions from...Ch. 4 - Prob. 4.20PCh. 4 - Answer true or false to the following statements...Ch. 4 - In each of the following three reaction coordinate...Ch. 4 - The acid-base chemistry reaction of barium...Ch. 4 - Unless under pressure, carbonic acid (H2CO3) in...Ch. 4 - Prob. 4.25PCh. 4 - Acetic acid, CH3COOH, is a weak organic acid, pKa...Ch. 4 - Benzoic acid, C6H5COOH (pKa 4.19), is only...Ch. 4 - Prob. 4.28PCh. 4 - One way to determine the predominant species at...Ch. 4 - Will acetylene react with sodium hydride according...Ch. 4 - Prob. 4.31PCh. 4 - For each equation, label the Lewis acid and the...Ch. 4 - Complete the equation for the reaction between...Ch. 4 - Each of these reactions can be written as a Lewis...Ch. 4 - The sec-butyl cation can react as both a...Ch. 4 - Prob. 4.36APCh. 4 - Prob. 4.37APCh. 4 - Prob. 4.38APCh. 4 - Explain why the hydronium ion, H3O+, is the...Ch. 4 - What is the strongest base that can exist in...Ch. 4 - Prob. 4.42APCh. 4 - Prob. 4.43APCh. 4 - Methyl isocyanate, CH3N=C=O, is used in the...Ch. 4 - Offer an explanation for the following...Ch. 4 - Prob. 4.46APCh. 4 - Alcohols (Chapter 10) are weak organic acids, pKa...Ch. 4 - As we shall see in Chapter 19, hydrogens on a...Ch. 4 - 2,4-Pentanedione is a considerably stronger acid...Ch. 4 - Write an equation for the acid-base reaction...Ch. 4 - Prob. 4.51APCh. 4 - Prob. 4.52APCh. 4 - Prob. 4.53APCh. 4 - Following is a structural formula for imidazole, a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Describe the acidity of different carboxylic acids and predict the products obtained when they react with strong bases.arrow_forwardHow to draw the line bond formula or lewis structure of a methyl ketone with the chemical formula C6H5C3H5O? Based on the results of the solubility tests, the compound can be grouped in VI, which is insoluble in water, 10% NaOH and 10% HCl but soluble in concentrated H2SO4. If the compound is water-insoluble but soluble in concentrated H2SO4, it is most probably an alcohol, aldehyde, anhydride, ester, ether, ketone, or an unsaturated hydrocarbon The functional group/class is identified to be Methyl Ketone, base on the results of the chemical tests on Table 2. CHEMICAL TEST OBSERVATIONS +(compound tested positive for the chemical reaction)/ otherwise (-) Molisch test turbid colorless solution - 2,4-DNP test formation of orange-yellow precipitates + Tollen’s test turbid colorless solution - Ninhydrin test clear pale-yellow solution - iodoform test clear pale-yellow solution +arrow_forwardIn the chemical reaction shown below, 0.0100 mol of 2-methyl-2-propanol is reacted with 0.0175 mol of ammonium bromide and 0.0350 mol of ammonium chloride in the presence of 0.100 mol of sulfuric acid. What are the products of this reaction and their respective ratio to one another? OH respectively) respectively) 2-methyl-2-propanol 2-bromo-2-methylpropane and 2-chloro-2-methylpropane (ratio: 1:2; NH₂*Cr NH₂ Br respectively) H₂SO4 H₂O 2-bromo-2-methylpropane and 2-chloro-2-methylpropane (ratio: 1:1; ? + ? respectively) 2-bromo-2-methylpropane and 2-chloro-2-methylpropane (ratio: 2:1; 1-bromo-2-methylpropane and 1-chloro-2-methylpropane (ratio: 1:2;arrow_forward
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardDescribe the trends in the physical properties of carboxylic acids, and contrast theirphysical properties with those of their salts.arrow_forwardWhat is the structure of the principal organic product formed in the reaction of 1-iodopropane with NaOH?arrow_forward
- Explain in detail how anthracene will mix with toluene.arrow_forwardThe normal pH range for blood plasma is 7.35–7.45. Under these conditions, would you expect the carboxyl group of lactic acid (pKa 3.08) to exist primarily as a carboxyl group or as a carboxylic anion? Explain.arrow_forward2-chloro-1-phenylpropane is reacted with ammonia, producing amphetamine, a widely misused stimulant. Write the chemical equation for the synthesis of amphetamine as described showing the structure of the compounds.arrow_forward
- What product is formed when the mixture of methyl-2-bromine-2-butane with isopropyl bromide and metallic sodium is heated? Write the chemical equation of the reactionarrow_forwardPrepare CH3COOC2H5, ethyl acetate, using ethyl alcohol. Write the chemical reaction. The pain reliever acetaminophen is produced by reacting 4-aminophenol with acetic anhydride. Outline a synthesis of acetaminophen from 4-aminophenol including any needed inorganic reagents.arrow_forwardWrite the chemical equation for a pentanoate ion acting as base when it reacts with hydrochloric acid (HCI). Which is formula of this rule of reaction?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License