
Concept explainers
Hydrocarbons like benzene are
phenols. This is an example of a general process in the body, in which an unwanted compound
(benzene) is converted to a more water-soluble derivative called a metabolite, so that it can be
excreted more readily from the body.
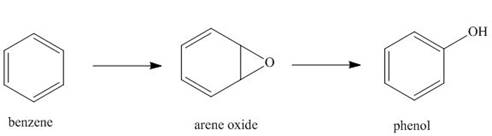
a. Classify each of these reactions as oxidation, reduction, or neither.
b. Explain why phenol is more water soluble than benzene. This means that phenol dissolves in urine, which is largely water, to a greater extent than benzene.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
EBK ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Organic Chemistry - Standalone book
General, Organic, & Biological Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry & Chemical Reactivity
Organic Chemistry As a Second Language: Second Semester Topics
Basic Chemistry (5th Edition)
- Alcohols and thiols can both be oxidized in a controlled way. What are the differences in the products?arrow_forwardThe REDOX reaction in the following is a. NaOH + HCI → NaCl + H₂O b. KOH →→ KCI + H₂O c. H2 + Cl2 →→→ HCI d. Ba(OH)₂ + HCI → BaCl₂ + H₂O O a. a O b. c O c. b O d. d The compound having Molecular formula C12H26 is a/an cycloalkane O alkyne O alkene O alkanearrow_forwardWrite equations for the complete combustion of each compound. propane (a bottled gas fuel) and octane (a typical hydrocarbon in gasoline).arrow_forward
- 17. lonic compounds dissolve in water through the phenomenon called. A. hydrogenation B. humidification C. hydration D. hallucination E. homogenizationarrow_forwardPetroleum ether is slowly oxidized by the action of peroxide. *arrow_forwardEthylene is oxidized in the presence of “X” as catalyst. What is the right pair of “X” and the product for an oxidation of ethylene a. A b. B c. C d. D e. Earrow_forward
- Classify the reaction type. a. oxidation (Benedict's) b. reduction (hydrogenation) c. acetal formation d. hemiacetal hydrolysis e. acetal hydrolysis f. mutarotation g. hemiacetal formationarrow_forwardWhat is the effect of carbon on the fuel combustion process? And what would happen if the carbon was high in the fuel ?arrow_forwardThe oxidation products of ethylene glycol (glycoaldehyde, glycolic acid, and oxalic acid) cause a variety of problems in the body. What happens to the oxalic acid? Which organs are damaged?arrow_forward
- The hydration of an alkene produces which organic functional group? a. alkene b. carboxylic acid c. aldehyde d. ether e. alcoholarrow_forwardHow many water molecules would be produced by the complete combustion of one straight chained butane molecule? A. 4 B. 5 C. 6 D. 8arrow_forward-Which substance has the greatest tendency to participate in a chemical reaction with small chain alcohol in an acidified chemical environment? a. Substance A b. Substance B c. Substance C d. Substance D e. Substance E -Which substance is expected to turn blue litmus paper into red color? a. Substance A b. Substance B c. Substance C d. Substance D e. Substance E -The physical properties of the substances are influenced by intermolecular forces of attraction. Which substance(s) are predominantly influenced by the intermolecular association of the molecules via hydrogen bonding? a.Substance A only b. Substance A and C c. Substance A, B, and C d.Substance A, B, C, and D -Aside from London dispersion forces, which substance do you expect dipole interactions to be effective? a. Substance A and B b. Substance B and D c. Substance A and C d. Substance B and E -Which substance will exhibit the lowest melting point? a. Substance A b. Substance B c. Substance C d. Substance D e. Substance E…arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

